Potent synergistic effect of IL-3 and TNF on matrix metalloproteinase 9 generation by human eosinophils
- PMID: 22321809
- PMCID: PMC3319644
- DOI: 10.1016/j.cyto.2012.01.009
Potent synergistic effect of IL-3 and TNF on matrix metalloproteinase 9 generation by human eosinophils
Abstract
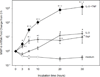
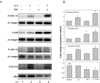
TNF (designated as TNF-α under previous nomenclature) is the preeminent activator of MMP-9 generation from a variety of cells including eosinophils. We have previously established that TNF strongly synergizes with IFN-γ and IL-4 for eosinophil synthesis of Th1- and Th2-type chemokines respectively. Thus, we sought to determine if TNF-induced synthesis of MMP-9 would be enhanced by the presence of Th1, Th2, or the eosinophil-associated common beta chain (βc) cytokines. Human blood eosinophils were cultured with TNF alone or in combination with either IFN-γ, IL-4, IL-3, IL-5, or GM-CSF. Concentrations and activities of MMP-9 in eosinophil culture supernates were measured by ELISA and gelatin zymography, mRNA transcription and stabilization by quantitative real-time PCR, and signaling events by immunoblotting and intracellular flow cytometric analysis. Individually, TNF, GM-CSF, or IL-3, but not IL-4 or IFN-γ, induced relatively small (<0.2 ng/ml) but statistically significant quantities of MMP-9. Remarkable synergistic synthesis of MMP-9 (ng/ml levels) occurred in response to TNF plus IL-3, GM-CSF or IL-5, in the order of IL-3>GM-CSF>IL-5. Zymography revealed that eosinophils release MMP-9 in its pro-form. Eosinophil stimulation with the combination of IL-3 plus TNF led to increased steady-state levels of MMP-9 mRNA, prolonged mRNA stabilization, and enhanced activation of ERK1/2 phosphorylation. Inhibition of NF-κB, MEK kinase, or p38 MAP kinase, but not JNK signaling pathways, diminished IL-3/TNF-induced MMP-9 mRNA and protein production. Thus, the synergistic regulation of eosinophil MMP-9 by IL-3 plus TNF likely involves cooperative interaction of multiple transcription factors downstream from ERK, p38, and NF-κB activation as well as post-transcriptional regulation of MMP-9 mRNA stabilization. Our data indicate that within microenvironments rich in βc-family cytokines and TNF, eosinophils are an important source of proMMP-9 and highlight a previously unrecognized role for synergistic interaction between TNF and βc-family cytokines, particularly IL-3, for proMMP-9 synthesis.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures







References
-
- Vempati P, Karagiannis ED, Popel AS. A biochemical model of matrix metalloproteinase 9 activation and inhibition. J Biol Chem. 2007;282:37585–37596. - PubMed
-
- Zitka O, Kukacka J, Krizkova S, Huska D, Adam V, Masarik M, et al. Matrix metalloproteinases. Curr Med Chem. 2010;17:3751–3768. - PubMed
-
- Hadler-Olsen E, Fadnes B, Sylte I, Uhlin-Hansen L, Winberg JO. Regulation of matrix metalloproteinase activity in health and disease. FEBS J. 2011;278:28–45. - PubMed
-
- Kelly EA, Jarjour NN. Role of MMPs in asthma. Curr Opin Pulm Med. 2003;9:28–33. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

