Phenolic compounds and antioxidant activities of Liriope muscari
- PMID: 22328078
- PMCID: PMC6268958
- DOI: 10.3390/molecules17021797
Phenolic compounds and antioxidant activities of Liriope muscari
Abstract
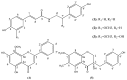
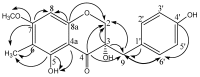
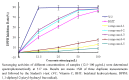
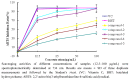
Five phenolic compounds, namely N-trans-coumaroyltyramine (1), N-trans-feruloyltyramine (2), N-trans-feruloyloctopamine (3), 5,7-dihydroxy-8-methoxyflavone (4) and (3S)3,5,4'-trihydroxy-7-methoxy-6-methylhomoisoflavanone (5), were isolated from the fibrous roots of Liriope muscari (Liliaceae). Compounds 2-5 were isolated for the first time from the Liriope genus. Their in vitro antioxidant activities were assessed by the DPPH and ABTS scavenging methods with microplate assays. The structure-activity relationships of compounds 1-3 are discussed.
Figures
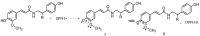
References
-
- Moure A., Cruz J.M., Franco D. Natural antioxidants fromresidual sources. Food Chem. 2001;72:145–171. doi: 10.1016/S0308-8146(00)00223-5. - DOI
-
- Oktay M., Gülçin İ., Küfrevioğlu Ö.İ. Determination of in vitro antioxidant activity of fennel (Foeniculum vulgare) seed extracts. LWT-Food Sci. Technol. 2003;36:263–271.
-
- Celik B., Lee J.H., Min D.B. Effects of light, oxygen and pH on the 2,2-diphenyl-1-picrylhydra-zyl (DPPH) method to evaluate antioxidants. J. Food Sci. 2003;68:487–490. doi: 10.1111/j.1365-2621.2003.tb05699.x. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

