β-catenin is a central mediator of pro-fibrotic Wnt signaling in systemic sclerosis
- PMID: 22328737
- PMCID: PMC3951949
- DOI: 10.1136/annrheumdis-2011-200568
β-catenin is a central mediator of pro-fibrotic Wnt signaling in systemic sclerosis
Abstract
Objectives: Pathologic fibroblast activation drives fibrosis of the skin and internal organs in patients with systemic sclerosis (SSc). β-catenin is an integral part of adherens junctions and a central component of canonical Wnt signaling. Here, the authors addressed the role of β-catenin in fibroblasts for the development of SSc dermal fibrosis.
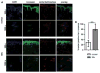
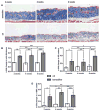
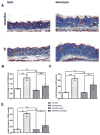
Methods: Nuclear accumulation of β-catenin in fibroblasts was assessed by triple staining for β-catenin, prolyl-4-hydroxylase-β and 4',6-diamidino-2-phenylindole (DAPI). The expression of Wnt proteins in the skin was analysed by real-time PCR and immunohistochemistry. Mice with fibroblast-specific stabilisation or fibroblast-specific depletion were used to evaluate the role of β-catenin in fibrosis.
Results: The auhors found significantly increased nuclear levels of β-catenin in fibroblasts in SSc skin compared to fibroblasts in the skin of healthy individuals. The accumulation of β-catenin resulted from increased expression of Wnt-1 and Wnt-10b in SSc. The authors further showed that the nuclear accumulation of β-catenin has direct implications for the development of fibrosis: Mice with fibroblast-specific stabilisation of β-catenin rapidly developed fibrosis within 2 weeks with dermal thickening, accumulation of collagen and differentiation of resting fibroblasts into myofibroblasts. By contrast, fibroblast-specific deletion of β-catenin significantly reduced bleomycin-induced dermal fibrosis.
Conclusions: The present study findings identify β-catenin as a key player of fibroblast activation and tissue fibrosis in SSc. Although further translational studies are necessary to test the efficacy and tolerability of β-catenin/Wnt inhibition in SSc, the present findings may have clinical implications, because selective inhibitors of β-catenin/Wnt signaling have recently entered clinical trials.
Conflict of interest statement
Figures

References
-
- Gabrielli A, Avvedimento EV, Krieg T. Scleroderma. N Engl J Med. 2009;360:1989–2003. - PubMed
-
- Chung L, Krishnan E, Chakravarty EF. Hospitalizations and mortality in systemic sclerosis: results from the Nationwide Inpatient Sample. Rheumatology (Oxford) 2007;46:1808–13. - PubMed
-
- van Amerongen R, Nusse R. Towards an integrated view of Wnt signaling in development. Development. 2009;136:3205–14. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

