Histone deacetylase 6 gates the synaptic action of acute stress in prefrontal cortex
- PMID: 22331421
- PMCID: PMC3413488
- DOI: 10.1113/jphysiol.2011.224907
Histone deacetylase 6 gates the synaptic action of acute stress in prefrontal cortex
Abstract
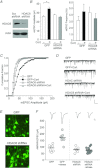
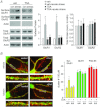
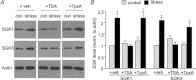
The prefrontal cortex (PFC), a region responsible for high-order cognitive functions, such as decision-making, attention and working memory, is highly influenced by stress and corticosteroid stress hormones. Recently it has been shown that acute stress affects PFC functions by potentiating glutamatergic transmission via a mechanism dependent on glucocorticoid receptor (GR) and its downstream target, serum and glucocorticoid-inducible kinase (SGK). To identify the key regulators of stress responses, we examined the role of histone deacetylase 6 (HDAC6), a unique member of the HDAC family that could regulate the GR chaperone protein heat shock protein 90 (HSP90), in the synaptic action of acute stress in PFC. We found that HDAC6 inhibition or knockdown blocked the enhancement of glutamatergic transmission and glutamate receptor trafficking by acute stress in vivo or corticosterone treatment in vitro. In addition, HDAC6 inhibition blocked the up-regulation of SGK in animals exposed to acute stress. HSP90 inhibition or knockdown produced a similar blockade of the acute stress-induced enhancement of glutamatergic signalling. These findings have identified HDAC6 as a key molecule gating the effects of acute stress on synaptic functions in the PFC.
Figures


Similar articles
-
HDAC6 regulates glucocorticoid receptor signaling in serotonin pathways with critical impact on stress resilience.J Neurosci. 2012 Mar 28;32(13):4400-16. doi: 10.1523/JNEUROSCI.5634-11.2012. J Neurosci. 2012. PMID: 22457490 Free PMC article.
-
Mechanisms for acute stress-induced enhancement of glutamatergic transmission and working memory.Mol Psychiatry. 2011 Feb;16(2):156-70. doi: 10.1038/mp.2010.50. Epub 2010 May 11. Mol Psychiatry. 2011. PMID: 20458323 Free PMC article.
-
Histone Modification of Nedd4 Ubiquitin Ligase Controls the Loss of AMPA Receptors and Cognitive Impairment Induced by Repeated Stress.J Neurosci. 2016 Feb 17;36(7):2119-30. doi: 10.1523/JNEUROSCI.3056-15.2016. J Neurosci. 2016. PMID: 26888924 Free PMC article.
-
Histone deacetylase 6 in health and disease.Epigenomics. 2015;7(1):103-18. doi: 10.2217/epi.14.69. Epigenomics. 2015. PMID: 25687470 Review.
-
Drugging the HDAC6-HSP90 interplay in malignant cells.Trends Pharmacol Sci. 2014 Oct;35(10):501-9. doi: 10.1016/j.tips.2014.08.001. Epub 2014 Sep 16. Trends Pharmacol Sci. 2014. PMID: 25234862 Review.
Cited by
-
Lysine Acetylation is an Important Post-Translational Modification that Modulates Heat Shock Response in the Sea Cucumber Apostichopus japonicus.Int J Mol Sci. 2019 Sep 9;20(18):4423. doi: 10.3390/ijms20184423. Int J Mol Sci. 2019. PMID: 31505730 Free PMC article.
-
The stressed synapse 2.0: pathophysiological mechanisms in stress-related neuropsychiatric disorders.Nat Rev Neurosci. 2022 Feb;23(2):86-103. doi: 10.1038/s41583-021-00540-x. Epub 2021 Dec 10. Nat Rev Neurosci. 2022. PMID: 34893785 Review.
-
CK1δ over-expressing mice display ADHD-like behaviors, frontostriatal neuronal abnormalities and altered expressions of ADHD-candidate genes.Mol Psychiatry. 2020 Dec;25(12):3322-3336. doi: 10.1038/s41380-018-0233-z. Epub 2018 Oct 19. Mol Psychiatry. 2020. PMID: 31363163 Free PMC article.
-
Neural deficits in a mouse model of PACS1 syndrome are corrected with PACS1- or HDAC6-targeting therapy.Nat Commun. 2023 Oct 17;14(1):6547. doi: 10.1038/s41467-023-42176-8. Nat Commun. 2023. PMID: 37848409 Free PMC article.
-
Corticosterone-mediated regulation and functions of miR-218-5p in rat brain.Sci Rep. 2022 Jan 7;12(1):194. doi: 10.1038/s41598-021-03863-y. Sci Rep. 2022. PMID: 34996981 Free PMC article.
References
-
- Aoyagi S, Archer TK. Modulating molecular chaperone Hsp90 functions through reversible acetylation. Trends Cell Biol. 2005;15:565–567. - PubMed
-
- Bali P, Pranpat M, Bradner J, Balasis M, Fiskus W, Guo F, Rocha K, Kumaraswamy S, Boyapalle S, Atadja P, Seto E, Bhalla K. Inhibition of histone deacetylase 6 acetylates and disrupts the chaperone function of heat shock protein 90: a novel basis for antileukemia activity of histone deacetylase inhibitors. J Biol Chem. 2005;280:26729–26734. - PubMed
-
- Cadepond F, Schweizer-Groyer G, Segard-Maurel I, Jibard N, Hollenberg SM, Giguère V, Evans RM, Baulieu EE. Heat shock protein 90 as a critical factor in maintaining glucocorticosteroid receptor in a nonfunctional state. J Biol Chem. 1991;266:5834–5841. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

