Spontaneous generation of rapidly transmissible prions in transgenic mice expressing wild-type bank vole prion protein
- PMID: 22331873
- PMCID: PMC3295307
- DOI: 10.1073/pnas.1121556109
Spontaneous generation of rapidly transmissible prions in transgenic mice expressing wild-type bank vole prion protein
Abstract
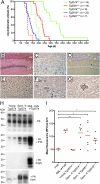
Currently, there are no animal models of the most common human prion disorder, sporadic Creutzfeldt-Jakob disease (CJD), in which prions are formed spontaneously from wild-type (WT) prion protein (PrP). Interestingly, bank voles (BV) exhibit an unprecedented promiscuity for diverse prion isolates, arguing that bank vole PrP (BVPrP) may be inherently prone to adopting misfolded conformations. Therefore, we constructed transgenic (Tg) mice expressing WT BVPrP. Tg(BVPrP) mice developed spontaneous CNS dysfunction between 108 and 340 d of age and recapitulated the hallmarks of prion disease, including spongiform degeneration, pronounced astrogliosis, and deposition of alternatively folded PrP in the brain. Brain homogenates of ill Tg(BVPrP) mice transmitted disease to Tg(BVPrP) mice in ∼35 d, to Tg mice overexpressing mouse PrP in under 100 d, and to WT mice in ∼185 d. Our studies demonstrate experimentally that WT PrP can spontaneously form infectious prions in vivo. Thus, Tg(BVPrP) mice may be useful for studying the spontaneous formation of prions, and thus may provide insight into the etiology of sporadic CJD.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Towards authentic transgenic mouse models of heritable PrP prion diseases.Acta Neuropathol. 2016 Oct;132(4):593-610. doi: 10.1007/s00401-016-1585-6. Epub 2016 Jun 28. Acta Neuropathol. 2016. PMID: 27350609 Free PMC article.
-
PrPC Governs Susceptibility to Prion Strains in Bank Vole, While Other Host Factors Modulate Strain Features.J Virol. 2016 Nov 14;90(23):10660-10669. doi: 10.1128/JVI.01592-16. Print 2016 Dec 1. J Virol. 2016. PMID: 27654300 Free PMC article.
-
Convergent generation of atypical prions in knockin mouse models of genetic prion disease.J Clin Invest. 2024 Aug 1;134(15):e176344. doi: 10.1172/JCI176344. J Clin Invest. 2024. PMID: 39087478 Free PMC article.
-
The utility of bank voles for studying prion disease.Prog Mol Biol Transl Sci. 2020;175:179-211. doi: 10.1016/bs.pmbts.2020.08.009. Epub 2020 Sep 8. Prog Mol Biol Transl Sci. 2020. PMID: 32958232 Review.
-
Prion protein transgenes and the neuropathology in prion diseases.Brain Pathol. 1995 Jan;5(1):77-89. doi: 10.1111/j.1750-3639.1995.tb00579.x. Brain Pathol. 1995. PMID: 7767493 Review.
Cited by
-
Creationism and evolutionism in prions.Am J Pathol. 2013 Mar;182(3):623-7. doi: 10.1016/j.ajpath.2012.12.016. Epub 2013 Feb 1. Am J Pathol. 2013. PMID: 23380581 Free PMC article.
-
A single protective polymorphism in the prion protein blocks cross-species prion replication in cultured cells.J Neurochem. 2023 Apr;165(2):230-245. doi: 10.1111/jnc.15739. Epub 2022 Dec 24. J Neurochem. 2023. PMID: 36511154 Free PMC article.
-
Cofactors facilitate bona fide prion misfolding in vitro but are not necessary for the infectivity of recombinant murine prions.PLoS Pathog. 2025 Jan 22;21(1):e1012890. doi: 10.1371/journal.ppat.1012890. eCollection 2025 Jan. PLoS Pathog. 2025. PMID: 39841704 Free PMC article.
-
Development of a new largely scalable in vitro prion propagation method for the production of infectious recombinant prions for high resolution structural studies.PLoS Pathog. 2019 Oct 23;15(10):e1008117. doi: 10.1371/journal.ppat.1008117. eCollection 2019 Oct. PLoS Pathog. 2019. PMID: 31644574 Free PMC article.
-
Selective vulnerability to neurodegenerative disease: the curious case of Prion Protein.Dis Model Mech. 2014 Jan;7(1):21-9. doi: 10.1242/dmm.012146. Dis Model Mech. 2014. PMID: 24396151 Free PMC article. Review.
References
-
- Prusiner SB. Novel proteinaceous infectious particles cause scrapie. Science. 1982;216:136–144. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P01 AG002132/AG/NIA NIH HHS/United States
- R37 AG031220/AG/NIA NIH HHS/United States
- NS041997/NS/NINDS NIH HHS/United States
- P01 AG010770/AG/NIA NIH HHS/United States
- CAPMC/ CIHR/Canada
- P01 AG021601/AG/NIA NIH HHS/United States
- AG021601/AG/NIA NIH HHS/United States
- R01 AI064709/AI/NIAID NIH HHS/United States
- AG031220/AG/NIA NIH HHS/United States
- AI064709/AI/NIAID NIH HHS/United States
- P01 NS041997/NS/NINDS NIH HHS/United States
- AG02132/AG/NIA NIH HHS/United States
- AG10770/AG/NIA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

