Catalytic site cysteines of thiol enzyme: sulfurtransferases
- PMID: 22332003
- PMCID: PMC3276061
- DOI: 10.4061/2011/709404
Catalytic site cysteines of thiol enzyme: sulfurtransferases
Abstract
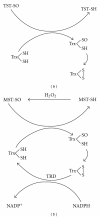
Thiol enzymes have single- or double-catalytic site cysteine residues and are redox active. Oxidoreductases and isomerases contain double-catalytic site cysteine residues, which are oxidized to a disulfide via a sulfenyl intermediate and reduced to a thiol or a thiolate. The redox changes of these enzymes are involved in their catalytic processes. On the other hand, transferases, and also some phosphatases and hydrolases, have a single-catalytic site cysteine residue. The cysteines are redox active, but their sulfenyl forms, which are inactive, are not well explained biologically. In particular, oxidized forms of sulfurtransferases, such as mercaptopyruvate sulfurtransferase and thiosulfate sulfurtransferase, are not reduced by reduced glutathione but by reduced thioredoxin. This paper focuses on why the catalytic site cysteine of sulfurtransferase is redox active.
Figures

Similar articles
-
Redox regulation of mammalian 3-mercaptopyruvate sulfurtransferase.Methods Enzymol. 2015;554:229-54. doi: 10.1016/bs.mie.2014.11.017. Epub 2015 Jan 20. Methods Enzymol. 2015. PMID: 25725525
-
Thioredoxin-dependent enzymatic activation of mercaptopyruvate sulfurtransferase. An intersubunit disulfide bond serves as a redox switch for activation.J Biol Chem. 2007 Jan 19;282(3):1561-9. doi: 10.1074/jbc.M605931200. Epub 2006 Nov 27. J Biol Chem. 2007. PMID: 17130129
-
Is novel signal transducer sulfur oxide involved in the redox cycle of persulfide at the catalytic site cysteine in a stable reaction intermediate of mercaptopyruvate sulfurtransferase?Antioxid Redox Signal. 2012 Apr 15;16(8):747-53. doi: 10.1089/ars.2011.4468. Epub 2012 Jan 11. Antioxid Redox Signal. 2012. PMID: 22149235
-
The mercaptopyruvate pathway in cysteine catabolism: a physiologic role and related disease of the multifunctional 3-mercaptopyruvate sulfurtransferase.Curr Med Chem. 2006;13(10):1219-30. doi: 10.2174/092986706776360914. Curr Med Chem. 2006. PMID: 16719781 Review.
-
A novel mercaptopyruvate sulfurtransferase thioredoxin-dependent redox-sensing molecular switch: a mechanism for the maintenance of cellular redox equilibrium.Mini Rev Med Chem. 2008 Jun;8(6):585-9. doi: 10.2174/138955708784534409. Mini Rev Med Chem. 2008. PMID: 18537713 Review.
Cited by
-
Biostimulation of Bacteria in Liquid Culture for Identification of New Antimicrobial Compounds.Pharmaceuticals (Basel). 2021 Nov 27;14(12):1232. doi: 10.3390/ph14121232. Pharmaceuticals (Basel). 2021. PMID: 34959632 Free PMC article.
-
Comparability of a three-dimensional structure in biopharmaceuticals using spectroscopic methods.J Anal Methods Chem. 2014;2014:950598. doi: 10.1155/2014/950598. Epub 2014 May 22. J Anal Methods Chem. 2014. PMID: 24963443 Free PMC article.
-
Sulfur Administration in Fe-S Cluster Homeostasis.Antioxidants (Basel). 2021 Oct 29;10(11):1738. doi: 10.3390/antiox10111738. Antioxidants (Basel). 2021. PMID: 34829609 Free PMC article. Review.
-
Cinnamaldehyde in flavored e-cigarette liquids temporarily suppresses bronchial epithelial cell ciliary motility by dysregulation of mitochondrial function.Am J Physiol Lung Cell Mol Physiol. 2019 Mar 1;316(3):L470-L486. doi: 10.1152/ajplung.00304.2018. Epub 2019 Jan 3. Am J Physiol Lung Cell Mol Physiol. 2019. PMID: 30604630 Free PMC article.
-
The cysteine proteome.Free Radic Biol Med. 2015 Jul;84:227-245. doi: 10.1016/j.freeradbiomed.2015.03.022. Epub 2015 Apr 3. Free Radic Biol Med. 2015. PMID: 25843657 Free PMC article. Review.
References
-
- Nagahara N, Matsumura T, Okamoto R, Kajihara Y. Protein cysteine modifications: (1) medicinal chemistry for proteomics. Current Medicinal Chemistry. 2009;16(33):4419–4444. - PubMed
-
- Nagahara N, Matsumura T, Okamoto R, Kajihara Y. Protein cysteine modifications: (2) reactivity specificity and topics of medicinal chemistry and protein engineering. Current Medicinal Chemistry. 2009;16(34):4490–4501. - PubMed
-
- Nagahara N. Intermolecular disulfide bond to modulate protein function as a redox-sensing switch. Amino Acids. In press. - PubMed
-
- Holmgren A. Glutathione-dependent synthesis of deoxyribonucleotides. Purification and characterization of glutaredoxin from Escherichia coli . Journal of Biological Chemistry. 1979;254(9):3664–3671. - PubMed
LinkOut - more resources
Full Text Sources

