Heme-copper terminal oxidase using both cytochrome c and ubiquinol as electron donors
- PMID: 22334648
- PMCID: PMC3295305
- DOI: 10.1073/pnas.1121040109
Heme-copper terminal oxidase using both cytochrome c and ubiquinol as electron donors
Abstract
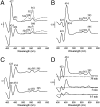
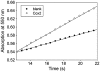
The cytochrome c oxidase Cox2 has been purified from native membranes of the hyperthermophilic eubacterium Aquifex aeolicus. It is a cytochrome ba(3) oxidase belonging to the family B of the heme-copper containing terminal oxidases. It consists of three subunits, subunit I (CoxA2, 63.9 kDa), subunit II (CoxB2, 16.8 kDa), and an additional subunit IIa of 5.2 kDa. Surprisingly it is able to oxidize both reduced cytochrome c and ubiquinol in a cyanide sensitive manner. Cox2 is part of a respiratory chain supercomplex. This supercomplex contains the fully assembled cytochrome bc(1) complex and Cox2. Although direct ubiquinol oxidation by Cox2 conserves less energy than ubiquinol oxidation by the cytochrome bc(1) complex followed by cytochrome c oxidation by a cytochrome c oxidase, ubiquinol oxidation by Cox2 is of advantage when all ubiquinone would be completely reduced to ubiquinol, e.g., by the sulfidequinone oxidoreductase, because the cytochrome bc(1) complex requires the presence of ubiquinone to function according to the Q-cycle mechanism. In the case that all ubiquinone has been reduced to ubiquinol its reoxidation by Cox2 will enable the cytochrome bc(1) complex to resume working.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


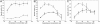
References
-
- Saraste M. Oxidative phosphorylation at the fin de siecle. Science. 1999;283:1488–1493. - PubMed
-
- Pereira MM, Santana M, Teixeira M. A novel scenario for the evolution of haem-copper oxygen reductases. Biochim Biophys Acta. 2001;1505:185–208. - PubMed
-
- Iwata S, Ostermeier C, Ludwig B, Michel H. Structure at 2.8 Å resolution of cytochrome c oxidase from Paracoccus denitrificans. Nature. 1995;376:660–669. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Research Materials

