Inhibition of delta-like ligand 4 induces luteal hypervascularization followed by functional and structural luteolysis in the primate ovary
- PMID: 22334711
- PMCID: PMC3413503
- DOI: 10.1210/en.2011-1688
Inhibition of delta-like ligand 4 induces luteal hypervascularization followed by functional and structural luteolysis in the primate ovary
Abstract
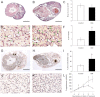
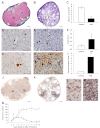
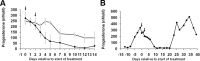
Using specific inhibitors established that angiogenesis in the ovarian follicle and corpus luteum is driven by vascular endothelial growth factor. Recently, it has been demonstrated that the Notch ligand, delta-like ligand 4 (Dll4) negatively regulates vascular endothelial growth factor-mediated vessel sprouting and branching. To investigate the role of Dll4 in regulation of the ovarian vasculature, we administered a neutralizing antibody to Dll4 to marmosets at the periovulatory period. The vasculature was examined on luteal d 3 or d 10: angiogenesis was determined by incorporation of bromodeoxyuridine, staining for CD31 and cell death by staining for activated caspase-3. Ovulatory progesterone rises were monitored to determine effects of treatment on luteal function and time to recover normal cycles in a separate group of animals. Additionally, animals were treated in the follicular or midluteal phase to determine effects of Dll4 inhibition on follicular development and luteal function. Controls were treated with human IgG (Fc). Corpora lutea from marmosets treated during the periovulatory period exhibited increased angiogenesis and increased vascular density on luteal d 3, but plasma progesterone was significantly suppressed. By luteal d 10, corpora lutea in treated ovaries were significantly reduced in size, with involution of luteal cells, increased cell death, and suppressed plasma progesterone concentrations. In contrast, initiation of anti-Dll4 treatment during the midluteal phase produced only a slight suppression of progesterone for the remainder of the cycle. Moreover, Dll4 inhibition had no appreciable effect on follicular development. These results show that Dll4 has a specific and critical role in the development of the normal luteal vasculature.
Figures
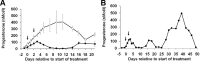
Similar articles
-
Administration of vascular endothelial growth factor Trap during the 'post-angiogenic' period of the luteal phase causes rapid functional luteolysis and selective endothelial cell death in the marmoset.Reproduction. 2006 Oct;132(4):589-600. doi: 10.1530/rep.1.01064. Reproduction. 2006. PMID: 17008470
-
Mid-luteal angiogenesis and function in the primate is dependent on vascular endothelial growth factor.J Endocrinol. 2001 Mar;168(3):409-16. doi: 10.1677/joe.0.1680409. J Endocrinol. 2001. PMID: 11241172
-
Suppression of luteal angiogenesis in the primate after neutralization of vascular endothelial growth factor.Endocrinology. 2000 Mar;141(3):995-1000. doi: 10.1210/endo.141.3.7369. Endocrinology. 2000. PMID: 10698175
-
SRB Reproduction, Fertility and Development Award Lecture 2008. Regulation and manipulation of angiogenesis in the ovary and endometrium.Reprod Fertil Dev. 2009;21(3):377-92. doi: 10.1071/rd08272. Reprod Fertil Dev. 2009. PMID: 19261215 Review.
-
Endocrine and local control of the primate corpus luteum.Reprod Biol. 2013 Dec;13(4):259-71. doi: 10.1016/j.repbio.2013.08.002. Epub 2013 Sep 14. Reprod Biol. 2013. PMID: 24287034 Free PMC article. Review.
Cited by
-
Roles of the Notch Signaling Pathway in Ovarian Functioning.Reprod Sci. 2021 Oct;28(10):2770-2778. doi: 10.1007/s43032-021-00610-6. Epub 2021 May 18. Reprod Sci. 2021. PMID: 34008156
-
The Role of Growth Hormone on Ovarian Functioning and Ovarian Angiogenesis.Front Endocrinol (Lausanne). 2019 Jul 16;10:450. doi: 10.3389/fendo.2019.00450. eCollection 2019. Front Endocrinol (Lausanne). 2019. PMID: 31379735 Free PMC article. Review.
-
Stage-dependent actions of antimüllerian hormone in regulating granulosa cell proliferation and follicular function in the primate ovary.F S Sci. 2020 Nov;1(2):161-171. doi: 10.1016/j.xfss.2020.10.005. Epub 2020 Oct 13. F S Sci. 2020. PMID: 34355206 Free PMC article.
-
The role of Notch signalling in ovarian angiogenesis.J Ovarian Res. 2017 Mar 11;10(1):13. doi: 10.1186/s13048-017-0308-5. J Ovarian Res. 2017. PMID: 28284219 Free PMC article. Review.
-
The Signaling Pathways Involved in Ovarian Follicle Development.Front Physiol. 2021 Sep 20;12:730196. doi: 10.3389/fphys.2021.730196. eCollection 2021. Front Physiol. 2021. PMID: 34646156 Free PMC article. Review.
References
-
- Reynolds LP, Killilea SD, Redmer DA. 1992. Angiogenesis in the female reproductive system. FASEB J 6:886–892 - PubMed
-
- Hazzard TM, Stouffer RL. 2000. Angiogenesis in ovarian follicular and luteal development. Baillieres Best Pract Res Clin Obstet Gynaecol 14:883–900 - PubMed
-
- Stocco C, Telleria C, Gibori G. 2007. The molecular control of corpus luteum formation, function, and regression. Endocr Rev 28:117–149 - PubMed
-
- Robinson RS, Woad KJ, Hammond AJ, Laird M, Hunter MG, Mann GE. 2009. Angiogenesis and vascular function in the ovary. Reproduction 138:869–881 - PubMed
-
- Fraser HM, Duncan WC. 2009. Regulation and manipulation of angiogenesis in the ovary and endometrium. Reprod Fertil Dev 21:377–392 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

