[A randomized clinical trial on the clinical efficacy and toxicities of single-agent paclitaxel liposome versus paclitaxel liposome plus oxaliplatin as first-line chemotherapy for advanced non-small cell lung cancer in elderly patients]
- PMID: 22336235
- PMCID: PMC6000255
- DOI: 10.3779/j.issn.1009-3419.2012.02.04
[A randomized clinical trial on the clinical efficacy and toxicities of single-agent paclitaxel liposome versus paclitaxel liposome plus oxaliplatin as first-line chemotherapy for advanced non-small cell lung cancer in elderly patients]
Abstract
Background and objective: The third generation single-agent drug has been recommended as a first-line chemotherapy for elderly patients with advanced non-small cell lung cancer (NSCLC). The aim of the current radomized trial is to compare the clinical efficacy and toxicities of single-agent paclitaxel liposome versus paclitaxel liposome plus oxaliplatin as a first-line chemotherapy for elderly patients.
Methods: Sixty-nine advanced NSCLC patients from July 2008 to August 2010, confirmed with pathology or cytology and had never received treatment, were randomly divided into two groups. The first group was given 135 mg/m² of single-agent paclitaxel liposome on day 1 of each cycle. The second group was given 135 mg/m² paclitaxel liposome plus 125 mg/m² oxaliplatin on day 1 of each cycle. One cycle is composed of 21 days. Efficacy and toxicities could be evaluated after two or more cycles.
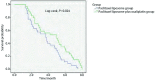
Results: No statistical differences were observed between the two groups in terms of efficacy (22.9% vs 35.3%, P=0.297), disease control rate (60.0% vs 70.6%, P=0.450), and 1-year survival rate (28.6% vs 41.2%, P=0.724). However, the group treated with paclitaxel liposome plus oxaliplatin had longer progression free survival (PFS) (5.0 months vs 3.5 months, P=0.024). In addition, the toxicities that occurred in the two groups were similar including leukocytopenia (P=0.808), thrombocytopenia (P>0.999), anemia (P=0.477), and nausea/vomiting (P=0.777). The number of neurotoxicity that occurred in the two groups were 33 and 3 (97.1% vs 8.6%, P<0.001), respectively. However, all were grade I-II.
Conclusions: The clinical efficacy of paclitaxel liposome plus oxaliplatin as a first-line chemotherapy for elderly patients with advanced NSCLC is more better than that of the single-agent paclitaxel liposome. It prolongs PFS and is safe for clinical use.
背景与目的: 目前推荐第三代药物单药治疗老年晚期非小细胞肺癌(non-small cell lung cancer, NSCLC),本研究旨在比较紫杉醇脂质体与紫杉醇脂质体联合奥沙利铂一线治疗老年晚期NSCLC的临床疗效及毒副作用。
方法: 2008年7月-2010年8月未经过治疗的经病理学或细胞学确诊的老年晚期NSCLC患者69例随机分成紫杉醇脂质体单药组(35例)和紫杉醇脂质体联合奥沙利铂组(34例),单药组给予紫杉醇脂质体135 mg/m2 d1;联合组给予紫杉醇脂质体135 mg/m2 d1+奥沙利铂125 mg/m2 d1,每21天重复,至少治疗2个周期,评价疗效和不良反应。
结果: 单药组与联合组相比,治疗有效率(22.9% vs 35.3%, P=0.297)、疾病控制率(60.0% vs 70.6%, P=0.450)和1年生存率(28.6% vs 41.2%, P=0.724)差异均无统计学意义,联合组的无疾病进展生存期(progression free survival, PFS)较单药组延长1.5个月(5.0个月 vs 3.5个月,P=0.024)。在毒副作用方面,两组白细胞减少(P=0.808)、血小板减少(P>0.999)、贫血(P=0.477)、恶心和呕吐的发生率(P=0.777)相当;两组发生神经毒性的患者分别为33例和3例(97.1% vs 8.6%, P<0.001),但均为Ⅰ度-Ⅱ度。
结论: 紫杉醇脂质体联合奥沙利铂用于一线治疗老年晚期NSCLC疗效略优于紫杉醇脂质体单药,能延长患者的PFS,临床应用安全性好。
Figures
Similar articles
-
[A randomized study of gemcitabine plus oxaliplatin versus gemcitabine plus cisplatin as the 1st line chemotherapy for advanced non-small cell lung cancer in elderly patients].Zhongguo Fei Ai Za Zhi. 2011 Jul;14(7):588-92. doi: 10.3779/j.issn.1009-3419.2011.07.05. Zhongguo Fei Ai Za Zhi. 2011. PMID: 21762628 Free PMC article. Clinical Trial. Chinese.
-
[A randomized controlled study of chemotherapy: etoposide combined with oxaliplatin or cisplatin regimens in the treatment of extensive-stage small cell lung cancer in elderly patients].Zhongguo Fei Ai Za Zhi. 2013 Jan;16(1):20-4. doi: 10.3779/j.issn.1009-3419.2013.01.04. Zhongguo Fei Ai Za Zhi. 2013. PMID: 23327869 Free PMC article. Clinical Trial. Chinese.
-
Nedaplatin or oxaliplatin combined with paclitaxel and docetaxel as first-line treatment for patients with advanced non-small cell lung cancer.Med Sci Monit. 2014 Dec 30;20:2830-6. doi: 10.12659/MSM.891318. Med Sci Monit. 2014. PMID: 25548985 Free PMC article.
-
Phase II study of carboplatin-paclitaxel combination chemotherapy in elderly patients with advanced non-small cell lung cancer.Jpn J Clin Oncol. 2005 Apr;35(4):188-94. doi: 10.1093/jjco/hyi059. Jpn J Clin Oncol. 2005. PMID: 15845567 Review.
-
Oxaliplatin in first-line therapy for advanced non-small-cell lung cancer.Clin Lung Cancer. 2010 Jan;11(1):18-24. doi: 10.3816/CLC.2010.n.003. Clin Lung Cancer. 2010. PMID: 20085863 Review.
Cited by
-
Assessing the effectiveness and safety of liposomal paclitaxel in combination with cisplatin as first-line chemotherapy for patients with advanced NSCLC with regional lymph-node metastasis: study protocol for a randomized controlled trial (PLC-GC trial).Trials. 2013 Feb 15;14:45. doi: 10.1186/1745-6215-14-45. Trials. 2013. PMID: 23413951 Free PMC article. Clinical Trial.
References
-
- Tu LL, Chen YS, Ding ZY, et al. Phase Ⅱ trail of paclitaxel liposome in elderly patients with non-small cell lung cancer. http://med.wanfangdata.com.cn/Paper/Detail/PeriodicalPaper_lczlxzz200904007 Chin Clin Oncol. 2009;14(4):314–318.
- 涂 玲莉, 陈 永顺, 丁 振宇, et al. 紫杉醇脂质体治疗老年非小细胞肺癌Ⅱ期临床研究. http://med.wanfangdata.com.cn/Paper/Detail/PeriodicalPaper_lczlxzz200904007 临床肿瘤学杂志. 2009;14(4):314–318.
-
- ES Santos, LE Raez, MF Rosado, et al. Efficacy and safety profile of oxaliplatin and docetaxel in patients with advanced non-small cell lung cancer (NSCLC) J Clin Oncol (Meeting Abstracts) 2006;24(18):17061.