Intermediate-term mortality and cardiac transplantation in infants with single-ventricle lesions: risk factors and their interaction with shunt type
- PMID: 22341427
- PMCID: PMC3359406
- DOI: 10.1016/j.jtcvs.2012.01.016
Intermediate-term mortality and cardiac transplantation in infants with single-ventricle lesions: risk factors and their interaction with shunt type
Abstract
Objective: The study objective was to identify factors associated with death and cardiac transplantation in infants undergoing the Norwood procedure and to determine differences in associations that might favor the modified Blalock-Taussig shunt or a right ventricle-to-pulmonary artery shunt.
Methods: We used competing risks methodology to analyze death without transplantation, cardiac transplantation, and survival without transplantation. Parametric time-to-event modeling and bootstrapping were used to identify independent predictors.
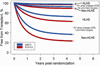
Results: Data from 549 subjects (follow-up, 2.7 ± 0.9 years) were analyzed. Mortality risk was characterized by early and constant phases; transplant was characterized by only a constant phase. Early phase factors associated with death included lower socioeconomic status (P = .01), obstructed pulmonary venous return (P < .001), smaller ascending aorta (P = .02), and anatomic subtype. Constant phase factors associated with death included genetic syndrome (P < .001) and lower gestational age (P < .001). The right ventricle-to-pulmonary artery shunt demonstrated better survival in the 51% of subjects who were full term with aortic atresia (P < .001). The modified Blalock-Taussig shunt was better among the 4% of subjects who were preterm with a patent aortic valve (P = .003). Lower pre-Norwood right ventricular fractional area change, pre-Norwood surgery, and anatomy other than hypoplastic left heart syndrome were independently associated with transplantation (all P < .03), but shunt type was not (P = .43).
Conclusions: Independent risk factors for intermediate-term mortality include lower socioeconomic status, anatomy, genetic syndrome, and lower gestational age. Term infants with aortic atresia benefited from a right ventricle-to-pulmonary artery shunt, and preterm infants with a patent aortic valve benefited from a modified Blalock-Taussig shunt. Right ventricular function and anatomy, but not shunt type, were associated with transplantation.
Trial registration: ClinicalTrials.gov NCT00115934.
Copyright © 2012 The American Association for Thoracic Surgery. All rights reserved.
Figures




References
-
- Diez Roux AV, Merkin SS, Arnett D, Chambless L, Massing M, Nieto FJ, et al. Neighborhood of residence and incidence of coronary heart disease. N Engl J Med. 2001 Jul 12;345(2):99–106. - PubMed
-
- Blackstone EH, Naftel DC, Turner ME. The decomposition of time-varying hazard Into phases, each Incorporating a separate stream of concomitant Information. Amer Stat Assoc. 1986;81:615–624.
Publication types
MeSH terms
Associated data
Grants and funding
- HL068288/HL/NHLBI NIH HHS/United States
- K23 HD061601/HD/NICHD NIH HHS/United States
- HL068285/HL/NHLBI NIH HHS/United States
- U01 HL068269/HL/NHLBI NIH HHS/United States
- U01 HL068279/HL/NHLBI NIH HHS/United States
- U01 HL068290/HL/NHLBI NIH HHS/United States
- U10 HL068270/HL/NHLBI NIH HHS/United States
- U01 HL068281/HL/NHLBI NIH HHS/United States
- U01 HL085057/HL/NHLBI NIH HHS/United States
- HL068279/HL/NHLBI NIH HHS/United States
- U01 HL068288/HL/NHLBI NIH HHS/United States
- U01 HL068270/HL/NHLBI NIH HHS/United States
- HL085057/HL/NHLBI NIH HHS/United States
- HL068281/HL/NHLBI NIH HHS/United States
- U01 HL068292/HL/NHLBI NIH HHS/United States
- HL068269/HL/NHLBI NIH HHS/United States
- HL068270/HL/NHLBI NIH HHS/United States
- HL068290/HL/NHLBI NIH HHS/United States
- U10 HL109673/HL/NHLBI NIH HHS/United States
- U01 HL068285/HL/NHLBI NIH HHS/United States
- HL068292/HL/NHLBI NIH HHS/United States

