Glycosphingolipids mediate pneumocystis cell wall β-glucan activation of the IL-23/IL-17 axis in human dendritic cells
- PMID: 22343219
- PMCID: PMC3402796
- DOI: 10.1165/rcmb.2011-0159OC
Glycosphingolipids mediate pneumocystis cell wall β-glucan activation of the IL-23/IL-17 axis in human dendritic cells
Abstract
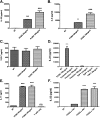
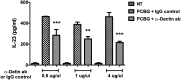
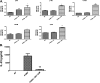
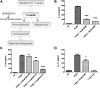
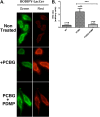
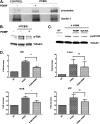
Pneumocystis species are opportunistic fungal organisms that cause severe pneumonia in immune-compromised hosts, with resultant high morbidity and mortality. Recent work indicates that IL-17 responses are important components of host defense against fungal pathogens. In the present study, we demonstrate that cell-surface β-glucan components of Pneumocystis (PCBG) stimulate human dendritic cells (DCs) to secrete IL-23 and IL-6. These cytokines are well established to stimulate a T helper-17 (Th17) phenotype. Accordingly, we further observe that PCBG-stimulated human DCs interact with lymphocytes to drive the secretion of IL-17 and IL-22, both Th17-produced cytokines. The activation of DCs was shown to involve the dectin-1 receptor with a downstream activation of the Syk kinase and subsequent translocation of both the canonical and noncanonical components of the NF-κB transcription factor family. Finally, we demonstrate that glycosphingolipid-rich microdomains of the plasma membrane participate in the activation of DCs by PCBG through the accumulation of lactosylceramide at the cell surface during stimulation with PCBG. These data strongly support the idea that the β-glucan surface components of Pneumocystis drive the activation of the IL-23/IL-17 axis during this infection, through a glycosphingolipid-initiated mechanism.
Figures

Similar articles
-
Pneumocystis cell wall beta-glucan stimulates calcium-dependent signaling of IL-8 secretion by human airway epithelial cells.Respir Res. 2010 Jul 13;11(1):95. doi: 10.1186/1465-9921-11-95. Respir Res. 2010. PMID: 20626862 Free PMC article.
-
Pneumocystis cell wall beta-glucans induce dendritic cell costimulatory molecule expression and inflammatory activation through a Fas-Fas ligand mechanism.J Immunol. 2006 Jul 1;177(1):459-67. doi: 10.4049/jimmunol.177.1.459. J Immunol. 2006. PMID: 16785543
-
Pneumocystis cell wall beta-glucans stimulate alveolar epithelial cell chemokine generation through nuclear factor-kappaB-dependent mechanisms.Am J Respir Cell Mol Biol. 2005 Jun;32(6):490-7. doi: 10.1165/rcmb.2004-0300OC. Epub 2005 Mar 3. Am J Respir Cell Mol Biol. 2005. PMID: 15746433 Free PMC article.
-
Current State of Carbohydrate Recognition and C-Type Lectin Receptors in Pneumocystis Innate Immunity.Front Immunol. 2021 Dec 16;12:798214. doi: 10.3389/fimmu.2021.798214. eCollection 2021. Front Immunol. 2021. PMID: 34975910 Free PMC article. Review.
-
CARD9 Syk-dependent and Raf-1 Syk-independent signaling pathways in target recognition of Candida albicans by Dectin-1.Eur J Clin Microbiol Infect Dis. 2011 Mar;30(3):303-5. doi: 10.1007/s10096-010-1103-z. Epub 2010 Nov 25. Eur J Clin Microbiol Infect Dis. 2011. PMID: 21108038 Review.
Cited by
-
IL-9 Deficiency Promotes Pulmonary Th17 Response in Murine Model of Pneumocystis Infection.Front Immunol. 2018 May 25;9:1118. doi: 10.3389/fimmu.2018.01118. eCollection 2018. Front Immunol. 2018. PMID: 29887863 Free PMC article.
-
New advances in understanding the host immune response to Pneumocystis.Curr Opin Microbiol. 2017 Dec;40:65-71. doi: 10.1016/j.mib.2017.10.019. Epub 2017 Nov 12. Curr Opin Microbiol. 2017. PMID: 29136537 Free PMC article. Review.
-
Bias of the Immune Response to Pneumocystis murina Does Not Alter the Ability of Neonatal Mice to Clear the Infection.J Fungi (Basel). 2021 Oct 2;7(10):827. doi: 10.3390/jof7100827. J Fungi (Basel). 2021. PMID: 34682248 Free PMC article.
-
Targeting β-glucans, vital components of the Pneumocystis cell wall.Front Immunol. 2023 Feb 9;14:1094464. doi: 10.3389/fimmu.2023.1094464. eCollection 2023. Front Immunol. 2023. PMID: 36845149 Free PMC article. Review.
-
Interactions of fungal pathogens with phagocytes.Nat Rev Microbiol. 2016 Mar;14(3):163-76. doi: 10.1038/nrmicro.2015.21. Epub 2016 Feb 8. Nat Rev Microbiol. 2016. PMID: 26853116 Review.
References
-
- Fischl MA, Dickinson GM, La Voie L. Safety and efficacy of sulfamethoxazole and trimethoprim chemoprophylaxis for Pneumocystis carinii pneumonia in aids. Jama 1988;259:1185–1189 - PubMed
-
- Masur H, Ognibene FP, Yarchoan R, Shelhamer JH, Baird BF, Travis W, Suffredini AF, Deyton L, Kovacs JA, Falloon J, et al. CD4 counts as predictors of opportunistic pneumonias in human immunodeficiency virus (HIV) infection. Ann Intern Med 1989;111:223–231 - PubMed
-
- Dungarwalla M, Marsh JC, Tooze JA, Lucas G, Ouwehand W, Pettengell R, Dearden CE, Gordon Smith EC, Elebute MO. Lack of clinical efficacy of rituximab in the treatment of autoimmune neutropenia and pure red cell aplasia: implications for their pathophysiology. Ann Hematol 2007;86:191–197 - PubMed
-
- Harigai M, Koike R, Miyasaka N. Pneumocystis pneumonia associated with infliximab in Japan. N Engl J Med 2007;357:1874–1876 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

