Standardizing immunophenotyping for the Human Immunology Project
- PMID: 22343568
- PMCID: PMC3409649
- DOI: 10.1038/nri3158
Standardizing immunophenotyping for the Human Immunology Project
Erratum in
- Nat Rev Immunol. 2012 Jun;12(6):471
Abstract
The heterogeneity in the healthy human immune system, and the immunological changes that portend various diseases, have been only partially described. Their comprehensive elucidation has been termed the 'Human Immunology Project'. The accurate measurement of variations in the human immune system requires precise and standardized assays to distinguish true biological changes from technical artefacts. Thus, to be successful, the Human Immunology Project will require standardized assays for immunophenotyping humans in health and disease. A major tool in this effort is flow cytometry, which remains highly variable with regard to sample handling, reagents, instrument setup and data analysis. In this Review, we outline the current state of standardization of flow cytometry assays and summarize the steps that are required to enable the Human Immunology Project.
Figures

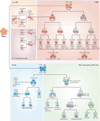

Comment in
-
Subjectivity and flow cytometric variability.Nat Rev Immunol. 2012 Apr 25;12(5):396; author reply 396. doi: 10.1038/nri3158-c1. Nat Rev Immunol. 2012. PMID: 22531327 No abstract available.
-
Standardization in flow cytometry: correct sample handling as a priority.Nat Rev Immunol. 2012 Dec;12(12):864. doi: 10.1038/nri3158-c3. Epub 2012 Nov 16. Nat Rev Immunol. 2012. PMID: 23154226 No abstract available.
References
-
- Perez OD, et al. Multiparameter analysis of intracellular phosphoepitopes in immunophenotyped cell populations by flow cytometry. Curr. Protoc. Cytom. 2005;32:6.20.1–6.20.22. - PubMed
-
- Maecker HT. In: Flow Cytometry Protocols. Hawley TS, Hawley RG, editors. Totowa, New Jersey: Humana Press; 2004. pp. 95–107.
-
- Parish CR, Glidden MH, Quah BJ, Warren HS. Use of the intracellular fluorescent dye CFSE to monitor lymphocyte migration and proliferation. Curr. Protoc. Immunol. 2009;84:4.9.1–4.9.13. - PubMed
-
- Betts MR, Koup RA. Detection of T-cell degranulation: CD107a and b. Methods Cell Biol. 2004;75:497–512. - PubMed
-
- Chattopadhyay PK, Yu J, Roederer M. A live-cell assay to detect antigen-specific CD4+ T cells with diverse cytokine profiles. Nature Med. 2005;11:1113–1117. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

