Gestures, vocalizations, and memory in language origins
- PMID: 22347184
- PMCID: PMC3269654
- DOI: 10.3389/fnevo.2012.00002
Gestures, vocalizations, and memory in language origins
Abstract
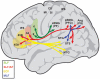
THIS ARTICLE DISCUSSES THE POSSIBLE HOMOLOGIES BETWEEN THE HUMAN LANGUAGE NETWORKS AND COMPARABLE AUDITORY PROJECTION SYSTEMS IN THE MACAQUE BRAIN, IN AN ATTEMPT TO RECONCILE TWO EXISTING VIEWS ON LANGUAGE EVOLUTION: one that emphasizes hand control and gestures, and the other that emphasizes auditory-vocal mechanisms. The capacity for language is based on relatively well defined neural substrates whose rudiments have been traced in the non-human primate brain. At its core, this circuit constitutes an auditory-vocal sensorimotor circuit with two main components, a "ventral pathway" connecting anterior auditory regions with anterior ventrolateral prefrontal areas, and a "dorsal pathway" connecting auditory areas with parietal areas and with posterior ventrolateral prefrontal areas via the arcuate fasciculus and the superior longitudinal fasciculus. In humans, the dorsal circuit is especially important for phonological processing and phonological working memory, capacities that are critical for language acquisition and for complex syntax processing. In the macaque, the homolog of the dorsal circuit overlaps with an inferior parietal-premotor network for hand and gesture selection that is under voluntary control, while vocalizations are largely fixed and involuntary. The recruitment of the dorsal component for vocalization behavior in the human lineage, together with a direct cortical control of the subcortical vocalizing system, are proposed to represent a fundamental innovation in human evolution, generating an inflection point that permitted the explosion of vocal language and human communication. In this context, vocal communication and gesturing have a common history in primate communication.
Keywords: arcuate fasciculus; broca’s area; inferior parietal lobe; mirror neurons; phonological loop; superior longitudinal fasciculus; working memory.
Figures

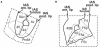
References
-
- Aboitiz F., Aboitiz S., García R. (2010). The phonological loop: a key innovation in human evolution. Curr. Anthropol. 51(Suppl. S1), S55–S65 10.1086/650525 - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

