Decrease in the oral bioavailability of dabigatran etexilate after co-medication with rifampicin
- PMID: 22348256
- PMCID: PMC3477350
- DOI: 10.1111/j.1365-2125.2012.04218.x
Decrease in the oral bioavailability of dabigatran etexilate after co-medication with rifampicin
Abstract
Aims: This study examined the effects of the CYP3A/P-glycoprotein inducer, rifampicin, on the pharmacokinetics of dabigatran following oral administration of the prodrug, dabigatran etexilate.
Methods: This was an open-label, fixed-sequence, four-period study in healthy volunteers. Subjects received a single dose of dabigatran etexilate 150 mg on day 1, rifampicin 600 mg once daily on days 2-8, and single doses of dabigatran etexilate on days 9, 16 and 23.
Results: Twenty-four subjects were treated, of whom 22 received all treatments. Relative to the reference (single dose of dabigatran etexilate alone; treatment A), administration of dabigatran etexilate following 7 days of rifampicin (treatment B) decreased the geometric mean (gMean) area under the concentration-time curve (AUC(0-∞)) and maximal plasma concentration (C(max)) of total dabigatran by 67 and 65.5%, respectively. The time to peak and the terminal half-life were not affected. The gMean ratio for the primary comparison (treatment B vs. treatment A) was 33.0% (90% confidence interval 26.5, 41.2%) for AUC(0-∞) and 34.5% (90% confidence interval 26.9, 44.1%) for C(max), indicating a significant effect on total dabigatran exposure (total pharmacologically active dabigatran represents the sum of nonconjugated dabigatran and dabigatran glucuronide). After a 7 day (treatment C) or 14 day washout (treatment D), the AUC(0-∞) and C(max) of dabigatran were reduced by 18 and 20%, and by 15 and 20%, respectively, compared with treatment A, which was considered not clinically relevant. The overall safety profile of all treatments was good.
Conclusions: Administration of rifampicin for 7 days resulted in a significant reduction in the bioavailability of dabigatran, which returned almost to baseline after 7 days washout.
© 2012 The Authors. British Journal of Clinical Pharmacology © 2012 The British Pharmacological Society.
Figures

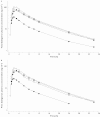
 ); Day 9 (period 2, test) (N = 24) (
); Day 9 (period 2, test) (N = 24) ( ); Day 16 (period 3, test) (N = 22) (
); Day 16 (period 3, test) (N = 22) ( ); Day 23 (period 4, test) (N = 22) (
); Day 23 (period 4, test) (N = 22) ( )
)
 ); gMean (n = 24/24/22/22) (
); gMean (n = 24/24/22/22) ( )
)Comment in
-
Rifampicin and dabigatran etexilate: a place for laboratory coagulation monitoring.Br J Clin Pharmacol. 2013 Feb;75(2):554-5. doi: 10.1111/j.1365-2125.2012.04408.x. Br J Clin Pharmacol. 2013. PMID: 23302069 Free PMC article. No abstract available.
-
Response to the letter by Dr Chin and co-workers.Br J Clin Pharmacol. 2013 Feb;75(2):556. doi: 10.1111/j.1365-2125.2012.04407.x. Br J Clin Pharmacol. 2013. PMID: 23302070 Free PMC article. No abstract available.
Similar articles
-
Pharmacokinetics and pharmacodynamics of the direct oral thrombin inhibitor dabigatran in healthy elderly subjects.Clin Pharmacokinet. 2008;47(1):47-59. doi: 10.2165/00003088-200847010-00005. Clin Pharmacokinet. 2008. PMID: 18076218 Clinical Trial.
-
Influence of renal impairment on the pharmacokinetics and pharmacodynamics of oral dabigatran etexilate: an open-label, parallel-group, single-centre study.Clin Pharmacokinet. 2010 Apr;49(4):259-68. doi: 10.2165/11318170-000000000-00000. Clin Pharmacokinet. 2010. PMID: 20214409 Clinical Trial.
-
Pharmacokinetic profile of the oral direct thrombin inhibitor dabigatran etexilate in healthy volunteers and patients undergoing total hip replacement.J Clin Pharmacol. 2005 May;45(5):555-63. doi: 10.1177/0091270005274550. J Clin Pharmacol. 2005. PMID: 15831779 Clinical Trial.
-
Clinical pharmacokinetics and pharmacodynamics of the oral direct thrombin inhibitor dabigatran etexilate.Clin Pharmacokinet. 2008;47(5):285-95. doi: 10.2165/00003088-200847050-00001. Clin Pharmacokinet. 2008. PMID: 18399711 Review.
-
Dabigatran: an oral novel potent reversible nonpeptide inhibitor of thrombin.Arterioscler Thromb Vasc Biol. 2010 Oct;30(10):1885-9. doi: 10.1161/ATVBAHA.110.203604. Epub 2010 Jul 29. Arterioscler Thromb Vasc Biol. 2010. PMID: 20671233 Review.
Cited by
-
Non-vitamin K Oral Anticoagulants and Anti-seizure Medications: A Retrospective Cohort Study.Front Neurol. 2021 Feb 26;11:588053. doi: 10.3389/fneur.2020.588053. eCollection 2020. Front Neurol. 2021. PMID: 33732201 Free PMC article.
-
Coexistence of Anticoagulant and Anti-vascular Calcification Activities in Kribbella sp. UTMC 267 Metabolites.Iran J Pharm Res. 2019 Winter;18(1):459-468. Iran J Pharm Res. 2019. PMID: 31089380 Free PMC article.
-
Cytochrome P450 3A Induction Predicts P-glycoprotein Induction; Part 1: Establishing Induction Relationships Using Ascending Dose Rifampin.Clin Pharmacol Ther. 2018 Dec;104(6):1182-1190. doi: 10.1002/cpt.1073. Epub 2018 Apr 19. Clin Pharmacol Ther. 2018. PMID: 29569723 Free PMC article. Clinical Trial.
-
Safety and effectiveness of anticoagulation with non-vitamin K antagonist oral anticoagulants and warfarin in patients on tuberculosis treatment.Sci Rep. 2023 Feb 4;13(1):2060. doi: 10.1038/s41598-023-29185-9. Sci Rep. 2023. PMID: 36739307 Free PMC article.
-
Clinical Management of Pharmacokinetic Drug Interactions with Direct Oral Anticoagulants (DOACs).Drugs. 2019 Oct;79(15):1625-1634. doi: 10.1007/s40265-019-01183-0. Drugs. 2019. PMID: 31440911 Review.
References
-
- Stangier J, Eriksson BI, Dahl OE, Ahnfelt L, Nehmiz G, Stähle H, Rathgen K, Svärd R. Pharmacokinetic profile of the oral direct thrombin inhibitor dabagitran etexilate in healthy volunteers and patients undergoing total hip replacement. J Clin Pharmacol. 2005;45:555–63. - PubMed
-
- Blech S, Ebner T, Ludwig-Schwellinger E, Stangier J, Roth W. The metabolism and disposition of the oral direct thrombin inhibitor, dabigatran, in humans. Drug Metab Dispos. 2008;36:386–99. - PubMed
-
- Stangier J. Clinical pharmacokinetics and pharmacodynamics of the oral direct thrombin inhibitor dabigatran etexilate. Clin Pharmacokinet. 2008;47:285–95. - PubMed
-
- Stangier J, Stähle H, Rathgen K, Fuhr R. Pharmacokinetics and pharmacodynamics of the direct oral thrombin inhibitor dabigatran in healthy elderly subjects. Clin Pharmacokinet. 2008;47:47–59. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

