Cellular force microscopy for in vivo measurements of plant tissue mechanics
- PMID: 22353572
- PMCID: PMC3343728
- DOI: 10.1104/pp.111.191460
Cellular force microscopy for in vivo measurements of plant tissue mechanics
Abstract
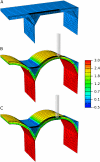
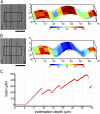
Although growth and morphogenesis are controlled by genetics, physical shape change in plant tissue results from a balance between cell wall loosening and intracellular pressure. Despite recent work demonstrating a role for mechanical signals in morphogenesis, precise measurement of mechanical properties at the individual cell level remains a technical challenge. To address this challenge, we have developed cellular force microscopy (CFM), which combines the versatility of classical microindentation techniques with the high automation and resolution approaching that of atomic force microscopy. CFM's large range of forces provides the possibility to map the apparent stiffness of both plasmolyzed and turgid tissue as well as to perform micropuncture of cells using very high stresses. CFM experiments reveal that, within a tissue, local stiffness measurements can vary with the level of turgor pressure in an unexpected way. Altogether, our results highlight the importance of detailed physically based simulations for the interpretation of microindentation results. CFM's ability to be used both to assess and manipulate tissue mechanics makes it a method of choice to unravel the feedbacks between mechanics, genetics, and morphogenesis.
Figures
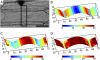
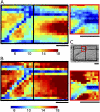
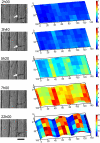
References
-
- Baskin TI. (2005) Anisotropic expansion of the plant cell wall. Annu Rev Cell Dev Biol 21: 203–222 - PubMed
-
- Blewett J, Burrows K, Thomas CA. (2000) Micromanipulation method to measure the mechanical properties of single tomato suspension cells. Biotechnol Lett 22: 1877–1883
-
- Bolduc JE, Lewis LJ, Aubin CE, Geitmann A. (2006) Finite-element analysis of geometrical factors in micro-indentation of pollen tubes. Biomech Model Mechanobiol 5: 227–236 - PubMed
-
- Burgert I. (2006) Exploring the micromechanical design of plant cell walls. Am J Bot 93: 1391–1401 - PubMed
-
- Cappella B, Dietler G. (1999) Force-distance curves by atomic force microscopy. Surf Sci Rep 34: 5–104
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

