Cdc42 and Rab8a are critical for intestinal stem cell division, survival, and differentiation in mice
- PMID: 22354172
- PMCID: PMC3287229
- DOI: 10.1172/JCI60282
Cdc42 and Rab8a are critical for intestinal stem cell division, survival, and differentiation in mice
Abstract
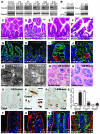
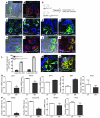
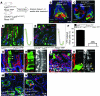
The constant self renewal and differentiation of adult intestinal stem cells maintains a functional intestinal mucosa for a lifetime. However, the molecular mechanisms that regulate intestinal stem cell division and epithelial homeostasis are largely undefined. We report here that the small GTPases Cdc42 and Rab8a are critical regulators of these processes in mice. Conditional ablation of Cdc42 in the mouse intestinal epithelium resulted in the formation of large intracellular vacuolar structures containing microvilli (microvillus inclusion bodies) in epithelial enterocytes, a phenotype reminiscent of human microvillus inclusion disease (MVID), a devastating congenital intestinal disorder that results in severe nutrient deprivation. Further analysis revealed that Cdc42-deficient stem cells had cell division defects, reduced capacity for clonal expansion and differentiation into Paneth cells, and increased apoptosis. Cdc42 deficiency impaired Rab8a activation and its association with multiple effectors, and prevented trafficking of Rab8a vesicles to the midbody. This impeded cytokinesis, triggering crypt apoptosis and disrupting epithelial morphogenesis. Rab8a was also required for Cdc42-GTP activity in the intestinal epithelium, where continued cell division takes place. Furthermore, mice haploinsufficient for both Cdc42 and Rab8a in the intestine demonstrated abnormal crypt morphogenesis and epithelial transporter physiology, further supporting their functional interaction. These data suggest that defects of the stem cell niche can cause MVID. This hypothesis represents a conceptual departure from the conventional view of this disease, which has focused on the affected enterocytes, and suggests stem cell-based approaches could be beneficial to infants with this often lethal condition.
Figures


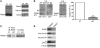

Similar articles
-
Cdc42 coordinates proliferation, polarity, migration, and differentiation of small intestinal epithelial cells in mice.Gastroenterology. 2013 Oct;145(4):808-19. doi: 10.1053/j.gastro.2013.06.021. Epub 2013 Jun 20. Gastroenterology. 2013. PMID: 23792201 Free PMC article.
-
RAB and RHO GTPases regulate intestinal crypt cell homeostasis and enterocyte function.Small GTPases. 2016 Apr 2;7(2):59-64. doi: 10.1080/21541248.2016.1159274. Epub 2016 May 4. Small GTPases. 2016. PMID: 27142493 Free PMC article. Review.
-
Rab8a vesicles regulate Wnt ligand delivery and Paneth cell maturation at the intestinal stem cell niche.Development. 2015 Jun 15;142(12):2147-62. doi: 10.1242/dev.121046. Epub 2015 May 26. Development. 2015. PMID: 26015543 Free PMC article.
-
Tuba Activates Cdc42 during Neuronal Polarization Downstream of the Small GTPase Rab8a.J Neurosci. 2021 Feb 24;41(8):1636-1649. doi: 10.1523/JNEUROSCI.0633-20.2020. Epub 2021 Jan 21. J Neurosci. 2021. PMID: 33478991 Free PMC article.
-
Regulation of intestinal stem cell fate specification.Sci China Life Sci. 2015 Jun;58(6):570-8. doi: 10.1007/s11427-015-4859-7. Epub 2015 May 8. Sci China Life Sci. 2015. PMID: 25951932 Review.
Cited by
-
Structural and functional hepatocyte polarity and liver disease.J Hepatol. 2015 Oct;63(4):1023-37. doi: 10.1016/j.jhep.2015.06.015. Epub 2015 Jun 24. J Hepatol. 2015. PMID: 26116792 Free PMC article. Review.
-
The role of cell division control protein 42 in tumor and non-tumor diseases: A systematic review.J Cancer. 2022 Jan 1;13(3):800-814. doi: 10.7150/jca.65415. eCollection 2022. J Cancer. 2022. PMID: 35154449 Free PMC article. Review.
-
Analysis of cell-specific transcriptional responses in human colon tissue using CIBERSORTx.Sci Rep. 2023 Oct 25;13(1):18281. doi: 10.1038/s41598-023-45582-6. Sci Rep. 2023. PMID: 37880448 Free PMC article.
-
The Endosomal Protein Endotubin Is Required for Enterocyte Differentiation.Cell Mol Gastroenterol Hepatol. 2017 Nov 15;5(2):145-156. doi: 10.1016/j.jcmgh.2017.11.001. eCollection 2018. Cell Mol Gastroenterol Hepatol. 2017. PMID: 29322087 Free PMC article.
-
An inducible mouse model for microvillus inclusion disease reveals a role for myosin Vb in apical and basolateral trafficking.Proc Natl Acad Sci U S A. 2015 Oct 6;112(40):12408-13. doi: 10.1073/pnas.1516672112. Epub 2015 Sep 21. Proc Natl Acad Sci U S A. 2015. PMID: 26392529 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

