Involvement of noradrenergic transmission in the PVN on CREB activation, TORC1 levels, and pituitary-adrenal axis activity during morphine withdrawal
- PMID: 22355339
- PMCID: PMC3280277
- DOI: 10.1371/journal.pone.0031119
Involvement of noradrenergic transmission in the PVN on CREB activation, TORC1 levels, and pituitary-adrenal axis activity during morphine withdrawal
Abstract
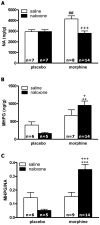
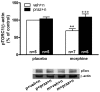
Experimental and clinical findings have shown that administration of adrenoceptor antagonists alleviated different aspects of drug withdrawal and dependence. The present study tested the hypothesis that changes in CREB activation and phosphorylated TORC1 levels in the hypothalamic paraventricular nucleus (PVN) after naloxone-precipitated morphine withdrawal as well as the HPA axis activity arises from α(1)- and/or β-adrenoceptor activation. The effects of morphine dependence and withdrawal on CREB phosphorylation (pCREB), phosphorylated TORC1 (pTORC1), and HPA axis response were measured by Western-blot, immunohistochemistry and radioimmunoassay in rats pretreated with prazosin (α(1)-adrenoceptor antagonist) or propranolol (β-adrenoceptor antagonist). In addition, the effects of morphine withdrawal on MHPG (the main NA metabolite at the central nervous system) and NA content and turnover were evaluated by HPLC. We found an increase in MHPG and NA turnover in morphine-withdrawn rats, which were accompanied by increased pCREB immunoreactivity and plasma corticosterone concentrations. Levels of the inactive form of TORC1 (pTORC1) were decreased during withdrawal. Prazosin but not propranolol blocked the rise in pCREB level and the decrease in pTORC1 immunoreactivity. In addition, the HPA axis response to morphine withdrawal was attenuated in prazosin-pretreated rats. Present results suggest that, during acute morphine withdrawal, NA may control the HPA axis activity through CREB activation at the PVN level. We concluded that the combined increase in CREB phosphorylation and decrease in pTORC1 levels might represent, in part, two of the mechanisms of CREB activation at the PVN during morphine withdrawal.
Conflict of interest statement
Figures


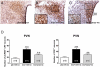

References
-
- Delfs JM, Zhu Y, Druhan JP, Aston-Jones G. Noradrenaline in the ventral forebrain is critical for opiate withdrawal-induced aversion. Nature. 2000;403:430–434. - PubMed
-
- Laorden ML, Fuertes G, González-Cuello A, Milanés MV. Changes in catecholaminergic pathways innervating paraventricular nucleus and pituitary-adrenal axis response during morphine dependence: implication of α1- and α2-adrenoceptors. J Pharmacol Exp Ther. 2000;293:578–584. - PubMed
-
- Harris GC, Aston-Jones G. Beta-adrenergic antagonists attenuate somatic and aversive signs of opiate withdrawal. Neuropsychopharmacology. 1993;9:303–311. - PubMed
-
- Sawchenko PE, Swanson LW. The organization of noradrenergic pathways from the brainstem to the paraventricular and supraoptic nuclei in the rat. Brain Res Rev. 1982;4:275–325. - PubMed
-
- Simpson TL, Saxon AJ, Meredith CW, Malte CA, McBride B, et al. A Pilot Trial of the Alpha-1 Adrenergic Antagonist, Prazosin, for Alcohol Dependence. Alcohol Clin Exp Res. 2009;33:255–263. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

