Herpes simplex virus 1 VP22 regulates translocation of multiple viral and cellular proteins and promotes neurovirulence
- PMID: 22357273
- PMCID: PMC3347367
- DOI: 10.1128/JVI.06913-11
Herpes simplex virus 1 VP22 regulates translocation of multiple viral and cellular proteins and promotes neurovirulence
Abstract
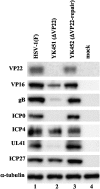
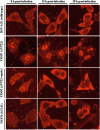
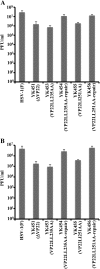
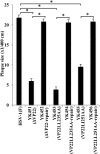
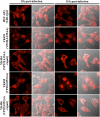
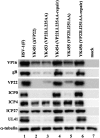
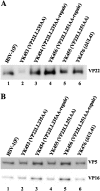
Herpes simplex virus 1 (HSV-1) protein VP22, encoded by the UL49 gene, is a major virion tegument protein. In the present study, we showed that VP22 was required for efficient redistribution of viral proteins VP16, VP26, ICP0, ICP4, and ICP27 and of cellular protein Hsc-70 to the cytoplasm of infected cells. We found that two dileucine motifs in VP22, at amino acids 235 and 236 and amino acids 251 and 252, were necessary for VP22 regulation of the proper cytoplasmic localization of these viral and cellular proteins. The dileucine motifs were also required for proper cytoplasmic localization of VP22 itself and for optimal expression of viral proteins VP16, VP22, ICP0, UL41, and glycoprotein B. Interestingly, a recombinant mutant virus with alanines substituted for the dileucines at amino acids 251 and 252 had a 50% lethal dose (LD(50)) for neurovirulence in mice following intracerebral inoculation about 10(3)-fold lower than the LD(50) of the repaired virus. Furthermore, the replication and spread of this mutant virus in the brains of mice following intracerebral inoculation were significantly impaired relative to those of the repaired virus. The ability of VP22 to regulate the localization and expression of various viral and cellular proteins, as shown in this study, was correlated with an increase in viral replication and neurovirulence in the experimental murine model. Thus, HSV-1 VP22 is a significant neurovirulence factor in vivo.
Figures






Similar articles
-
Alphaherpesvirus Major Tegument Protein VP22: Its Precise Function in the Viral Life Cycle.Front Microbiol. 2020 Aug 7;11:1908. doi: 10.3389/fmicb.2020.01908. eCollection 2020. Front Microbiol. 2020. PMID: 32849477 Free PMC article. Review.
-
Quantitative Evaluation of Protein Heterogeneity within Herpes Simplex Virus 1 Particles.J Virol. 2017 Apr 28;91(10):e00320-17. doi: 10.1128/JVI.00320-17. Print 2017 May 15. J Virol. 2017. PMID: 28275191 Free PMC article.
-
Multiple Posttranscriptional Strategies To Regulate the Herpes Simplex Virus 1 vhs Endoribonuclease.J Virol. 2018 Aug 16;92(17):e00818-18. doi: 10.1128/JVI.00818-18. Print 2018 Sep 1. J Virol. 2018. PMID: 29925667 Free PMC article.
-
A network of protein interactions around the herpes simplex virus tegument protein VP22.J Virol. 2012 Dec;86(23):12971-82. doi: 10.1128/JVI.01913-12. Epub 2012 Sep 19. J Virol. 2012. PMID: 22993164 Free PMC article.
-
Characterization of a UL49-null mutant: VP22 of herpes simplex virus type 1 facilitates viral spread in cultured cells and the mouse cornea.J Virol. 2006 Sep;80(17):8664-75. doi: 10.1128/JVI.00498-06. J Virol. 2006. PMID: 16912314 Free PMC article.
Cited by
-
Functional comparison of herpes simplex virus 1 (HSV-1) and HSV-2 ICP27 homologs reveals a role for ICP27 in virion release.J Virol. 2015 Mar;89(5):2892-905. doi: 10.1128/JVI.02994-14. Epub 2014 Dec 24. J Virol. 2015. PMID: 25540385 Free PMC article.
-
Alphaherpesvirus Major Tegument Protein VP22: Its Precise Function in the Viral Life Cycle.Front Microbiol. 2020 Aug 7;11:1908. doi: 10.3389/fmicb.2020.01908. eCollection 2020. Front Microbiol. 2020. PMID: 32849477 Free PMC article. Review.
-
Host factors associated with either VP16 or VP16-induced complex differentially affect HSV-1 lytic infection.Rev Med Virol. 2022 Nov;32(6):e2394. doi: 10.1002/rmv.2394. Epub 2022 Sep 7. Rev Med Virol. 2022. PMID: 36069169 Free PMC article. Review.
-
Phosphorylation of a herpes simplex virus 1 dUTPase by a viral protein kinase, Us3, dictates viral pathogenicity in the central nervous system but not at the periphery.J Virol. 2014 Mar;88(5):2775-85. doi: 10.1128/JVI.03300-13. Epub 2013 Dec 18. J Virol. 2014. PMID: 24352467 Free PMC article.
-
Characterization of Elements Regulating the Nuclear-to-Cytoplasmic Translocation of ICP0 in Late Herpes Simplex Virus 1 Infection.J Virol. 2018 Jan 2;92(2):e01673-17. doi: 10.1128/JVI.01673-17. Print 2018 Jan 15. J Virol. 2018. PMID: 29093084 Free PMC article.
References
-
- Asai R, Ohno T, Kato A, Kawaguchi Y. 2007. Identification of proteins directly phosphorylated by UL13 protein kinase from herpes simplex virus 1. Microbes Infect. 9:1434–1438 - PubMed
-
- Blaho JA, Mitchell C, Roizman B. 1994. An amino acid sequence shared by the herpes simplex virus 1 alpha regulatory proteins 0, 4, 22, and 27 predicts the nucleotidylylation of the UL21, UL31, UL47, and UL49 gene products. J. Biol. Chem. 269:17401–17410 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

