2,3,7,8-tetrachlorodibenzo-p-dioxin induces transcriptional activity of the human polymorphic hs1,2 enhancer of the 3'Igh regulatory region
- PMID: 22357631
- PMCID: PMC3311717
- DOI: 10.4049/jimmunol.1101111
2,3,7,8-tetrachlorodibenzo-p-dioxin induces transcriptional activity of the human polymorphic hs1,2 enhancer of the 3'Igh regulatory region
Abstract
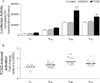
2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) is an environmental toxicant known to inhibit Ab secretion and Ig expression. Inhibition of Ig expression may be partially mediated through repression of the 3'Igh regulatory region (3'IghRR). TCDD inhibits mouse 3'IghRR activation and induces aryl hydrocarbon receptor binding to dioxin response elements within the 3'IghRR enhancers hs1,2 and hs4. The human hs1,2 enhancer (hu-hs1,2) is polymorphic as the result of the presence of one to four invariant sequences (ISs), which have been correlated with several autoimmune diseases. The IS also contains a dioxin response element core motif. Therefore, the objective was to determine whether hu-hs1,2 activity is sensitive to TCDD. Using a mouse B cell line (CH12.LX), we compared the effects of TCDD on mouse hs1,2 versus hu-hs1,2 activity. TCDD inhibited mouse hs1,2 similarly to the mouse 3'IghRR. In contrast, hu-hs1,2 was activated by TCDD, and antagonist studies supported an aryl hydrocarbon receptor-dependent activation, which was replicated in a human B cell line (IM-9). Absence of Pax5 binding sites is a major difference between the human and mouse hs1,2 sequence. Insertion of the high-affinity Pax5 site in hu-hs1,2 markedly blunted reporter activity but did not alter TCDD's effect (i.e., no shift from activation to inhibition). Additionally, deletional analysis demonstrated a significant IS contribution to hu-hs1,2 basal activity, but TCDD-induced activity was not strictly IS number dependent. Taken together, our results suggest that hu-hs1,2 is a significant target of TCDD and support species differences in hs1,2 regulation. Therefore, sensitivity of hu-hs1,2 to chemical-induced modulation may influence the occurrence and/or severity of human diseases associated with hu-hs1,2.
Figures








References
-
- Pinaud E, Khamlichi AA, Le Morvan C, Drouet M, Nalesso V, Le Bert M, Cogne M. Localization of the 3' IgH locus elements that effect long-distance regulation of class switch recombination. Immunity. 2001;15:187–199. - PubMed
-
- Vincent-Fabert C, Fiancette R, Pinaud E, Truffinet V, Cogne N, Cogne M, Denizot Y. Genomic deletion of the whole IgH 3' regulatory region (hs3a, hs1,2, hs3b, hs4) dramatically affects class switch recombination and Ig secretion to all isotypes. Blood. 2010;116:1895–1898. - PubMed
-
- Sulentic CEW, Holsapple MP, Kaminski NE. Putative link between transcriptional regulation of IgM expression by 2,3,7,8-tetrachlorodibenzo-p-dioxin and the aryl hydrocarbon receptor/dioxin-responsive enhancer signaling pathway. J. Pharmacol. Exp. Ther. 2000;295:705–716. - PubMed
-
- Sulentic CEW, Kang JS, Na YJ, Kaminski NE. Interactions at a dioxin responsive element (DRE) and an overlapping kappaB site within the hs4 domain of the 3'alpha immunoglobulin heavy chain enhancer. Toxicology. 2004;200:235–246. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

