The ubiquitin-proteasome system meets angiogenesis
- PMID: 22357635
- PMCID: PMC3297735
- DOI: 10.1158/1535-7163.MCT-11-0555
The ubiquitin-proteasome system meets angiogenesis
Abstract
A strict physiological balance between endogenous proangiogenic and antiangiogenic factors controls endothelial cell functions, such that endothelial cell growth is normally restrained. However, in pathologic angiogenesis, a shift occurs in the balance of regulators, favoring endothelial growth. Much of the control of angiogenic events is instigated through hypoxia-induced VEGF expression. The ubiquitin-proteasome system (UPS) plays a central role in fine-tuning the functions of core proangiogenic proteins, including VEGF, VEGFR-2, angiogenic signaling proteins (e.g., the PLCγ1 and PI3 kinase/AKT pathways), and other non-VEGF angiogenic pathways. The emerging mechanisms by which ubiquitin modification of angiogenic proteins control angiogenesis involve both proteolytic and nonproteolytic functions. Here, I review recent advances that link the UPS to regulation of angiogenesis and highlight the potential therapeutic value of the UPS in angiogenesis-associated diseases.
Figures



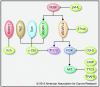
References
-
- Chung AS, Lee J, Ferrara N. Targeting the tumour vasculature: insights from physiological angiogenesis. Nat Rev Cancer. 2010;10:505–14. - PubMed
-
- Jain RK. Normalization of tumor vasculature: an emerging concept in anti-angiogenic therapy. Science. 2005;307(5706):58–62. - PubMed
-
- Afzal A, Shaw LC, Ljubimov AV, Boulton ME, Segal MS, Grant MB. Retinal and choroidal microangiopathies: therapeutic opportunities. Microvasc Res. 2007;74(2-3):131–44. - PubMed
-
- Brown MM, Brown GC, Stein JD, Roth Z, Campanella J, Beauchamp GR. Age-related macular degeneration: economic burden and value based medicine analysis. Can. J. Ophthalmol. 2005;40:277–287. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

