Mycobacterium tuberculosis ClpP1 and ClpP2 function together in protein degradation and are required for viability in vitro and during infection
- PMID: 22359499
- PMCID: PMC3280978
- DOI: 10.1371/journal.ppat.1002511
Mycobacterium tuberculosis ClpP1 and ClpP2 function together in protein degradation and are required for viability in vitro and during infection
Abstract
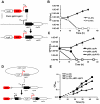
In most bacteria, Clp protease is a conserved, non-essential serine protease that regulates the response to various stresses. Mycobacteria, including Mycobacterium tuberculosis (Mtb) and Mycobacterium smegmatis, unlike most well studied prokaryotes, encode two ClpP homologs, ClpP1 and ClpP2, in a single operon. Here we demonstrate that the two proteins form a mixed complex (ClpP1P2) in mycobacteria. Using two different approaches, promoter replacement, and a novel system of inducible protein degradation, leading to inducible expression of clpP1 and clpP2, we demonstrate that both genes are essential for growth and that a marked depletion of either one results in rapid bacterial death. ClpP1P2 protease appears important in degrading missense and prematurely terminated peptides, as partial depletion of ClpP2 reduced growth specifically in the presence of antibiotics that increase errors in translation. We further show that the ClpP1P2 protease is required for the degradation of proteins tagged with the SsrA motif, a tag co-translationally added to incomplete protein products. Using active site mutants of ClpP1 and ClpP2, we show that the activity of each subunit is required for proteolysis, for normal growth of Mtb in vitro and during infection of mice. These observations suggest that the Clp protease plays an unusual and essential role in Mtb and may serve as an ideal target for antimycobacterial therapy.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



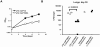
References
-
- Ingmer H, Brøndsted L. Proteases in bacterial pathogenesis. Res Microbiol. 2009;160:704–710. doi: 10.1016/j.resmic.2009.08.017. - DOI - PubMed
-
- Goldberg AL. Protein degradation and protection against misfolded or damaged proteins. Nature. 2003;426:895–899. doi: 10.1038/nature02263. - DOI - PubMed
-
- Glickman MH, Ciechanover A. The ubiquitin-proteasome proteolytic pathway: destruction for the sake of construction. Physiol Rev. 2002;82:373–428. doi: 10.1152/physrev.00027.2001. - DOI - PubMed
-
- Frees D, Savijoki K, Varmanen P, Ingmer H. Clp ATPases and ClpP proteolytic complexes regulate vital biological processes in low GC, Gram-positive bacteria. Mol Microbiol. 2007;63:1285–1295. doi: 10.1111/j.1365-2958.2007.05598.x. - DOI - PubMed
-
- Burns KE, Pearce MJ, Darwin KH. Prokaryotic ubiquitin-like protein provides a two-part degron to Mycobacterium proteasome substrates. J Bacteriol. 2010;192:2933–2935. doi: 10.1128/JB.01639-09. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

