A plasma biomarker signature of immune activation in HIV patients on antiretroviral therapy
- PMID: 22363505
- PMCID: PMC3281899
- DOI: 10.1371/journal.pone.0030881
A plasma biomarker signature of immune activation in HIV patients on antiretroviral therapy
Abstract
Background: Immune activation is a strong predictor of disease progression in HIV infection. Combinatorial plasma biomarker signatures that represent surrogate markers of immune activation in both viremic and aviremic HIV patients on combination antiretroviral therapy (cART) have not been defined. Here, we identify a plasma inflammatory biomarker signature that distinguishes between both viremic and aviremic HIV patients on cART and healthy controls and examine relationships of this signature to markers of disease progression.
Methods: Multiplex profiling and ELISA were used to detect 15 cytokines/chemokines, soluble IL-2R (sIL-2R), and soluble CD14 (sCD14) in plasma from 57 HIV patients with CD4 nadir <300 cells/µl and 29 healthy controls. Supervised and unsupervised analyses were used to identify biomarkers explaining variance between groups defined by HIV status or drug abuse. Relationships between biomarkers and disease markers were examined by Spearman correlation.
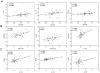
Results: The majority (91%) of HIV subjects were on cART, with 38% having undetectable viral loads (VL). Hierarchical clustering identified a biomarker cluster in plasma consisting of two interferon-stimulated gene products (CXCL9 and CXCL10), T cell activation marker (sIL-2R), and monocyte activation marker (sCD14) that distinguished both viremic and aviremic HIV patients on cART from controls (p<0.0001) and were top-ranked in variables important in projection plots. IL-12 and CCL4 were also elevated in viremic and aviremic patients compared to controls (p<0.05). IL-12 correlated with IFNα, IFNγ, CXCL9, and sIL-2R (p<0.05). CXCL10 correlated positively with plasma VL and percentage of CD16+ monocytes, and inversely with CD4 count (p = 0.001, <0.0001, and 0.04, respectively).
Conclusion: A plasma inflammatory biomarker signature consisting of CXCL9, CXCL10, sIL-2R, and sCD14 may be useful as a surrogate marker to monitor immune activation in both viremic and aviremic HIV patients on cART during disease progression and therapeutic responses.
Conflict of interest statement
Figures




References
-
- d'Ettorre G, Paiardini M, Ceccarelli G, Silvestri G, Vullo V. HIV-Associated Immune Activation: From Bench to Bedside. AIDS Res Hum Retroviruses 2011 - PubMed
-
- Ortiz AM, Silvestri G. Immunopathogenesis of AIDS. Curr Infect Dis Rep. 2009;11:239–245. - PubMed
-
- Appay V, Sauce D. Immune activation and inflammation in HIV-1 infection: causes and consequences. J Pathol. 2008;214:231–241. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- U01 MH083507/MH/NIMH NIH HHS/United States
- R24 MH059724/MH/NIMH NIH HHS/United States
- U01 MH083500/MH/NIMH NIH HHS/United States
- R24 NS045491/NS/NINDS NIH HHS/United States
- R24 NS038841/NS/NINDS NIH HHS/United States
- R24 NS45491/NS/NINDS NIH HHS/United States
- DP1 DA028994/DA/NIDA NIH HHS/United States
- U01 MH083501/MH/NIMH NIH HHS/United States
- U01 MH083506/MH/NIMH NIH HHS/United States
- UL1 RR029887/RR/NCRR NIH HHS/United States
- U01MH083545/MH/NIMH NIH HHS/United States
- 5U01MH083500/MH/NIMH NIH HHS/United States
- R24MH59724/MH/NIMH NIH HHS/United States
- CAPMC/ CIHR/Canada
- R24 MH059745/MH/NIMH NIH HHS/United States
- R24MH59745/MH/NIMH NIH HHS/United States
- N01 MH032002/MH/NIMH NIH HHS/United States
- DA28994/DA/NIDA NIH HHS/United States
- R01 DA030985/DA/NIDA NIH HHS/United States
- U01MH083501/MH/NIMH NIH HHS/United States
- U01MH083506/MH/NIMH NIH HHS/United States
- U01 MH083545/MH/NIMH NIH HHS/United States
- N01 MH022005/MH/NIMH NIH HHS/United States
- U01MH083507/MH/NIMH NIH HHS/United States
- P30 CA006516/CA/NCI NIH HHS/United States
- P30 AI060354/AI/NIAID NIH HHS/United States
- DA30985/DA/NIDA NIH HHS/United States
- NS38841/NS/NINDS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

