Novel covalently linked insulin dimer engineered to investigate the function of insulin dimerization
- PMID: 22363506
- PMCID: PMC3281904
- DOI: 10.1371/journal.pone.0030882
Novel covalently linked insulin dimer engineered to investigate the function of insulin dimerization
Abstract
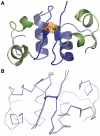
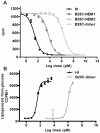
An ingenious system evolved to facilitate insulin binding to the insulin receptor as a monomer and at the same time ensure sufficient stability of insulin during storage. Insulin dimer is the cornerstone of this system. Insulin dimer is relatively weak, which ensures dissociation into monomers in the circulation, and it is stabilized by hexamer formation in the presence of zinc ions during storage in the pancreatic β-cell. Due to the transient nature of insulin dimer, direct investigation of this important form is inherently difficult. To address the relationship between insulin oligomerization and insulin stability and function, we engineered a covalently linked insulin dimer in which two monomers were linked by a disulfide bond. The structure of this covalent dimer was identical to the self-association dimer of human insulin. Importantly, this covalent dimer was capable of further oligomerization to form the structural equivalent of the classical hexamer. The covalently linked dimer neither bound to the insulin receptor, nor induced a metabolic response in vitro. However, it was extremely thermodynamically stable and did not form amyloid fibrils when subjected to mechanical stress, underlining the importance of oligomerization for insulin stability.
Conflict of interest statement
Figures


Similar articles
-
Structure, Aggregation, and Activity of a Covalent Insulin Dimer Formed During Storage of Neutral Formulation of Human Insulin.J Pharm Sci. 2016 Apr;105(4):1376-86. doi: 10.1016/j.xphs.2016.01.003. Epub 2016 Feb 23. J Pharm Sci. 2016. PMID: 26921119
-
Structure of an insulin dimer in an orthorhombic crystal: the structure analysis of a human insulin mutant (B9 Ser-->Glu).Acta Crystallogr D Biol Crystallogr. 1999 Sep;55(Pt 9):1524-32. doi: 10.1107/s0907444999008562. Acta Crystallogr D Biol Crystallogr. 1999. PMID: 10489447
-
Non-equivalent role of inter- and intramolecular hydrogen bonds in the insulin dimer interface.J Biol Chem. 2011 Oct 21;286(42):36968-77. doi: 10.1074/jbc.M111.265249. Epub 2011 Aug 31. J Biol Chem. 2011. PMID: 21880708 Free PMC article.
-
The road to the first, fully active and more stable human insulin variant with an additional disulfide bond.J Pept Sci. 2015 Nov;21(11):797-806. doi: 10.1002/psc.2822. Epub 2015 Sep 18. J Pept Sci. 2015. PMID: 26382042 Review.
-
Zinc-ligand interactions modulate assembly and stability of the insulin hexamer -- a review.Biometals. 2005 Aug;18(4):295-303. doi: 10.1007/s10534-005-3685-y. Biometals. 2005. PMID: 16158220 Review.
Cited by
-
4S-Hydroxylation of Insulin at ProB28 Accelerates Hexamer Dissociation and Delays Fibrillation.J Am Chem Soc. 2017 Jun 28;139(25):8384-8387. doi: 10.1021/jacs.7b00794. Epub 2017 Jun 20. J Am Chem Soc. 2017. PMID: 28598606 Free PMC article.
-
Total Chemical Synthesis of Palmitoyl-Conjugated Insulin.ACS Omega. 2023 Apr 6;8(15):13715-13720. doi: 10.1021/acsomega.2c07918. eCollection 2023 Apr 18. ACS Omega. 2023. PMID: 37091377 Free PMC article.
-
Insulin analog with additional disulfide bond has increased stability and preserved activity.Protein Sci. 2013 Mar;22(3):296-305. doi: 10.1002/pro.2211. Epub 2013 Jan 17. Protein Sci. 2013. PMID: 23281053 Free PMC article.
-
IgE clonality and aggregation of insulin affect IgE-mediated activation of sensitized basophils.J Allergy Clin Immunol Glob. 2025 May 28;4(3):100502. doi: 10.1016/j.jacig.2025.100502. eCollection 2025 Aug. J Allergy Clin Immunol Glob. 2025. PMID: 40583959 Free PMC article.
-
Long-term glycemic control and prevention of diabetes complications in vivo using oleic acid-grafted-chitosan‑zinc-insulin complexes incorporated in thermosensitive copolymer.J Control Release. 2020 Jul 10;323:161-178. doi: 10.1016/j.jconrel.2020.04.012. Epub 2020 Apr 10. J Control Release. 2020. PMID: 32283211 Free PMC article.
References
-
- Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med. 1998;15:539–553. - PubMed
-
- Baker EN, Blundell TL, Cutfield JF, Cutfield SM, Dodson EJ, et al. The structure of 2Zn pig insulin crystals at 1.5 A resolution. Philos Trans R Soc Lond B Biol Sci. 1988;319:369–456. - PubMed
-
- Steiner DF. The Banting Memorial Lecture 1976. Insulin today. Diabetes. 1977;26:322–340. - PubMed
-
- Dodson G, Steiner D. The role of assembly in insulin's biosynthesis. Curr Opin Struct Biol. 1998;8:189–194. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

