Zinc induces caspase-dependent mitochondrial pathway of the programmed cell death in haemocytes of Drosophila melanogaster
- PMID: 22367497
- PMCID: PMC3349848
- DOI: 10.1007/s10534-012-9530-1
Zinc induces caspase-dependent mitochondrial pathway of the programmed cell death in haemocytes of Drosophila melanogaster
Abstract
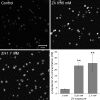
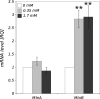
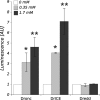
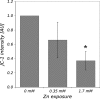
Zinc is an essential trace element in cells. However, its high level in cytoplasm promotes activation of stress signaling pathways and may lead to cell death. In the present study we used Drosophila melanogaster blood cells (haemocytes), obtained from the third instar larvae, to study the effects of high concentrations of Zn(2+) on programmed cell death (PCD). We analyzed the activity of caspases, the level of caspase inhibitor protein DIAP1 and metallothioneins, as well as calcium concentrations and activity of mitochondria in haemocytes exposed to 0.35 and 1.7 mM concentrations of Zn. The obtained results showed that rapid increase of [Zn(2+)]( i ) in the cytoplasm up-regulates metallothionein MtnB but not MtnA gene expression in cells treated with Zn(2+) in both concentrations. Excess of Zn(2+) also induced activation of the initiator caspase Dronc, associated with the mitochondrial pathway of PCD, and the effector caspase DrICE. In turn, the activity of receptor-regulated Dredd caspase was not changed. The level of DIAP1 decreased significantly in haemocytes in the presence of high Zn(2+) concentration in comparison to untreated cells. Moreover, mitochondrial membrane potential was significantly decreased after exposure to Zn ions. These results indicate that high concentration of Zn(2+) in the cytoplasm of haemocytes induces PCD via a mitochondrial pathway and that caspases play a pivotal role in this process.
Figures

Similar articles
-
Effects of zinc on programmed cell death of Musca domestica and Drosophila melanogaster blood cells.J Insect Physiol. 2010 Apr;56(4):383-90. doi: 10.1016/j.jinsphys.2009.11.010. Epub 2009 Dec 2. J Insect Physiol. 2010. PMID: 19941868
-
Drosophila caspases involved in developmentally regulated programmed cell death of peptidergic neurons during early metamorphosis.J Comp Neurol. 2011 Jan 1;519(1):34-48. doi: 10.1002/cne.22498. J Comp Neurol. 2011. PMID: 21120926
-
Cell survival and proliferation in Drosophila S2 cells following apoptotic stress in the absence of the APAF-1 homolog, ARK, or downstream caspases.Apoptosis. 2006 Apr;11(4):497-507. doi: 10.1007/s10495-006-5341-6. Apoptosis. 2006. PMID: 16532275
-
Drosophila caspases as guardians of host-microbe interactions.Cell Death Differ. 2023 Feb;30(2):227-236. doi: 10.1038/s41418-022-01038-4. Epub 2022 Jul 9. Cell Death Differ. 2023. PMID: 35810247 Free PMC article. Review.
-
Apoptosis in Drosophila: which role for mitochondria?Apoptosis. 2016 Mar;21(3):239-51. doi: 10.1007/s10495-015-1209-y. Apoptosis. 2016. PMID: 26679112 Review.
Cited by
-
A recessive X-linked mutation causes a threefold reduction of total body zinc accumulation in Drosophila melanogaster laboratory strains.FEBS Open Bio. 2013 Jul 24;3:302-4. doi: 10.1016/j.fob.2013.07.003. eCollection 2013. FEBS Open Bio. 2013. PMID: 23951551 Free PMC article.
-
Tracing heavy metals in 'swine manure - maggot - chicken' production chain.Sci Rep. 2017 Aug 21;7(1):8417. doi: 10.1038/s41598-017-07317-2. Sci Rep. 2017. PMID: 28827547 Free PMC article.
-
Anatomy and Physiology of the Digestive Tract of Drosophila melanogaster.Genetics. 2018 Oct;210(2):357-396. doi: 10.1534/genetics.118.300224. Genetics. 2018. PMID: 30287514 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

