DetoxiProt: an integrated database for detoxification proteins
- PMID: 22369658
- PMCID: PMC3333179
- DOI: 10.1186/1471-2164-12-S3-S2
DetoxiProt: an integrated database for detoxification proteins
Abstract
Background: Detoxification proteins are a class of proteins for degradation and/or elimination of endogenous and exogenous toxins or medicines, as well as reactive oxygen species (ROS) produced by these materials. Most of these proteins are generated as a response to the stimulation of toxins or medicines. They are essential for the clearance of harmful substances and for maintenance of physiological balance in organisms. Thus, it is important to collect and integrate information on detoxification proteins.
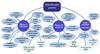
Results: To store, retrieve and analyze the information related to their features and functions, we developed the DetoxiProt, a comprehensive database for annotation of these proteins. This database provides detailed introductions about different classes of the detoxification proteins. Extensive annotations of these proteins, including sequences, structures, features, inducers, inhibitors, substrates, chromosomal location, functional domains as well as physiological-biochemical properties were generated. Furthermore, pre-computed BLAST results, multiple sequence alignments and evolutionary trees for detoxification proteins are also provided for evolutionary study of conserved function and pathways. The current version of DetoxiProt contains 5956 protein entries distributed in 628 organisms. An easy to use web interface was designed, so that annotations about each detoxification protein can be retrieved by browsing with a specific method or by searching with different criteria.
Conclusions: DetoxiProt provides an effective and efficient way of accessing the detoxification protein sequences and other high-quality information. This database would be a valuable source for toxicologists, pharmacologists and medicinal chemists. DetoxiProt database is freely available at http://lifecenter.sgst.cn/detoxiprot/.
Figures


Similar articles
-
PDTD: a web-accessible protein database for drug target identification.BMC Bioinformatics. 2008 Feb 19;9:104. doi: 10.1186/1471-2105-9-104. BMC Bioinformatics. 2008. PMID: 18282303 Free PMC article.
-
DBMLoc: a Database of proteins with multiple subcellular localizations.BMC Bioinformatics. 2008 Feb 28;9:127. doi: 10.1186/1471-2105-9-127. BMC Bioinformatics. 2008. PMID: 18304364 Free PMC article.
-
UniSave: the UniProtKB sequence/annotation version database.Bioinformatics. 2006 May 15;22(10):1284-5. doi: 10.1093/bioinformatics/btl105. Epub 2006 Mar 21. Bioinformatics. 2006. PMID: 16551660
-
Biomolecular interaction network database.Brief Bioinform. 2005 Jun;6(2):194-8. doi: 10.1093/bib/6.2.194. Brief Bioinform. 2005. PMID: 15975228 Review.
-
Filling and mining the reactive metabolite target protein database.Chem Biol Interact. 2009 Apr 15;179(1):38-44. doi: 10.1016/j.cbi.2008.08.016. Epub 2008 Sep 6. Chem Biol Interact. 2009. PMID: 18823962 Free PMC article. Review.
Cited by
-
Integrated genome-wide association and transcriptomic studies reveal genetic architecture of bulb storability of plentiful garlic germplasm resources.Hortic Res. 2024 Sep 16;11(12):uhae260. doi: 10.1093/hr/uhae260. eCollection 2024 Dec. Hortic Res. 2024. PMID: 39664692 Free PMC article.
-
Oxidative cleavage of polysaccharides by a termite-derived superoxide dismutase boosts the degradation of biomass by glycoside hydrolases.Green Chem. 2022 May 12;24(12):4845-4858. doi: 10.1039/d1gc04519a. eCollection 2022 Jun 20. Green Chem. 2022. PMID: 35813357 Free PMC article.
-
Identification of novel candidate gene associated with early heading on chromosome 5B in wheat mutant jg1489.Mol Breed. 2025 Jun 18;45(7):57. doi: 10.1007/s11032-025-01580-2. eCollection 2025 Jul. Mol Breed. 2025. PMID: 40547848
-
Critical controllability analysis of directed biological networks using efficient graph reduction.Sci Rep. 2017 Oct 30;7(1):14361. doi: 10.1038/s41598-017-14334-8. Sci Rep. 2017. PMID: 29084972 Free PMC article.
-
SeqSNP-Based Targeted GBS Provides Insight into the Genetic Relationships among Global Collections of Brassica rapa ssp. oleifera (Turnip Rape).Genes (Basel). 2024 Sep 10;15(9):1187. doi: 10.3390/genes15091187. Genes (Basel). 2024. PMID: 39336778 Free PMC article.
References
-
- Limòn-Pacheco J, Gonsebatt ME. The role of antioxidants and antioxidant-related enzymes in protective responses to environmentally induced oxidative stress. Mutat Res. 2009;674(1-2):137–147. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

