Importance of electrostatic polarizability in calculating cysteine acidity constants and copper(I) binding energy of Bacillus subtilis CopZ
- PMID: 22370900
- PMCID: PMC3306534
- DOI: 10.1002/jcc.22944
Importance of electrostatic polarizability in calculating cysteine acidity constants and copper(I) binding energy of Bacillus subtilis CopZ
Abstract
CopZ is a copper chaperone from Bacillus subtilis. It is an important part of Cu(I) trafficking. We have calculated pK(a) values for the CXXC motif of this protein, which is responsible for the Cu(I) binding, and the Cu(I) binding constants. Polarizable and fixed-charges formalisms were used, and solvation parameters for the both models have been refitted. We had to partially redevelop parameters for the protonated and deprotonated cysteine residues. We have discovered that the polarizable force field (PFF) is qualitatively superior and allows a uniformly better level of energetic results. The PFF pK(a) values for cysteine are within about 0.8-2.8 pH units of the experimental data, while the fixed-charges OPLS formalism yields errors of up to tens of units. The PFF magnitude of the copper binding energy is about 10 kcal/mol or 50% higher than the experimental value, while the using the refitted OPLS parameters leads to an overall positive binding energy, thus predicting no thermodynamically stable complex. At the same time, the agreement of the polarizable S···Cu(I) distances with the experimental results is within 0.08 Å range, and the nonpolarizable calculations lead to an error of about 0.4 Å. Moreover, the accuracy of the PFF has been achieved without any explicit fitting to either pK(a) or CopZ···Cu(I) binding energies. We believe that this makes our polarizable technique a choice method in reproducing protein-copper binding and further supports the notion that explicit treatment of electrostatic polarization is crucial in many biologically relevant studies, especially ion binding and transport.
Copyright © 2012 Wiley Periodicals, Inc.
Figures
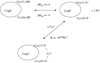

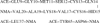
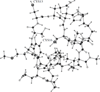
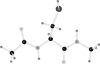


Similar articles
-
Electrostatic polarization is crucial in reproducing Cu(I) interaction energies and hydration.J Phys Chem B. 2011 Aug 25;115(33):10079-85. doi: 10.1021/jp2051933. Epub 2011 Jul 28. J Phys Chem B. 2011. PMID: 21761909 Free PMC article.
-
Tuning of copper-loop flexibility in Bacillus subtilis CopZ copper chaperone: role of conserved residues.J Phys Chem B. 2009 Feb 19;113(7):1919-32. doi: 10.1021/jp807594q. J Phys Chem B. 2009. PMID: 19170606
-
High Cu(I) and low proton affinities of the CXXC motif of Bacillus subtilis CopZ.Biochem J. 2008 Aug 1;413(3):459-65. doi: 10.1042/BJ20080467. Biochem J. 2008. PMID: 18419582
-
Structure and dynamics of Cu(I) binding in copper chaperones Atox1 and CopZ: a computer simulation study.J Phys Chem B. 2008 Apr 17;112(15):4583-93. doi: 10.1021/jp711787x. Epub 2008 Mar 25. J Phys Chem B. 2008. PMID: 18361527
-
Mechanistic insights into Cu(I) cluster transfer between the chaperone CopZ and its cognate Cu(I)-transporting P-type ATPase, CopA.Biochem J. 2009 Dec 10;424(3):347-56. doi: 10.1042/BJ20091079. Biochem J. 2009. PMID: 19751213
Cited by
-
Metal Ion Modeling Using Classical Mechanics.Chem Rev. 2017 Feb 8;117(3):1564-1686. doi: 10.1021/acs.chemrev.6b00440. Epub 2017 Jan 3. Chem Rev. 2017. PMID: 28045509 Free PMC article. Review.
-
Polarizable simulations with second order interaction model (POSSIM) force field: developing parameters for protein side-chain analogues.J Comput Chem. 2013 May 30;34(14):1241-50. doi: 10.1002/jcc.23248. Epub 2013 Feb 19. J Comput Chem. 2013. PMID: 23420678 Free PMC article.
-
Membrane Anchoring and Ion-Entry Dynamics in P-type ATPase Copper Transport.Biophys J. 2016 Dec 6;111(11):2417-2429. doi: 10.1016/j.bpj.2016.10.020. Biophys J. 2016. PMID: 27926843 Free PMC article.
-
POSSIM: Parameterizing Complete Second-Order Polarizable Force Field for Proteins.J Chem Theory Comput. 2014 Nov 11;10(11):4896-4910. doi: 10.1021/ct500243k. Epub 2014 Oct 14. J Chem Theory Comput. 2014. PMID: 25400518 Free PMC article.
-
Developing multisite empirical force field models for Pt(II) and cisplatin.J Comput Chem. 2017 Jan 30;38(3):161-168. doi: 10.1002/jcc.24665. Epub 2016 Nov 11. J Comput Chem. 2017. PMID: 27859392 Free PMC article.
References
-
-
See for example: Zhou L, Singleton C, Le Brun NE. Biochem. J. 2008;413:459–465. Badarau A, Dennison C. J. Am. Chem. Soc. 2011;133:2983–2988. Waldon KJ, Robinson NJ. Nat. Rev. Microbiol. 2009;7:25. Robinson NJ, Winge DR. Annu. Rev. Biochem. 2010;79:537–562. Banci L, Bertini I, Ciofi-Baffoni S, Kozyreva T, Zovo K, Palumaa P. Nature. 2010;465 645-U145. Banci L, Bertini I, Cantini F, Ciofi-Baffoni S. Cell. Mol. Life Sci. 2010;67:2563–2589. Bertini I, Cavallaro G, McGreevy KS. Coord. Chem. Rev. 2010;254:506–524.
-
-
- MacDermaid CM, Kaminski GA. J. Phys. Chem. B. 2007;111:9036–9044. - PubMed
-
- Kaminski GA. J. Phys. Chem. B. 2005;109:5884–5890. - PubMed
-
- Caldwell JW, Kollman PA. J. Am. Chem. Soc. 1995;117:4177–4178.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

