In vitro phosphorylation of the focal adhesion targeting domain of focal adhesion kinase by Src kinase
- PMID: 22372511
- PMCID: PMC3332079
- DOI: 10.1021/bi300123a
In vitro phosphorylation of the focal adhesion targeting domain of focal adhesion kinase by Src kinase
Abstract
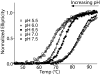
Focal adhesion kinase (FAK), a key regulator of cell adhesion and migration, is overexpressed in many types of cancer. The C-terminal focal adhesion targeting (FAT) domain of FAK is necessary for proper localization of FAK to focal adhesions and subsequent activation. Phosphorylation of Y926 in the FAT domain by the tyrosine kinase Src has been shown to promote metastasis and invasion in vivo by linking the FAT domain to the MAPK pathway via its interaction with growth factor receptor-bound protein 2. Several groups have reported that inherent conformational dynamics in the FAT domain likely regulate phosphorylation of Y926; however, what regulates these dynamics is unknown. In this paper, we demonstrate that there are two sites of in vitro Src-mediated phosphorylation in the FAT domain: Y926, which has been shown to affect FAK function in vivo, and Y1008, which has no known biological role. The phosphorylation of these two tyrosine residues is pH-dependent, but this does not reflect the pH dependence of Src kinase activity. Circular dichroism and nuclear magnetic resonance data indicate that the stability and conformational dynamics of the FAT domain are sensitive to changes in pH over a physiological pH range. In particular, regions of the FAT domain previously shown to regulate phosphorylation of Y926 as well as regions near Y1008 show pH-dependent dynamics on the microsecond to millisecond time scale.
Figures




Similar articles
-
Evidence for in vivo phosphorylation of the Grb2 SH2-domain binding site on focal adhesion kinase by Src-family protein-tyrosine kinases.Mol Cell Biol. 1996 Oct;16(10):5623-33. doi: 10.1128/MCB.16.10.5623. Mol Cell Biol. 1996. PMID: 8816475 Free PMC article.
-
Identification of Src-specific phosphorylation site on focal adhesion kinase: dissection of the role of Src SH2 and catalytic functions and their consequences for tumor cell behavior.Cancer Res. 2005 Feb 15;65(4):1335-42. doi: 10.1158/0008-5472.CAN-04-1949. Cancer Res. 2005. PMID: 15735019
-
Complex formation with focal adhesion kinase: A mechanism to regulate activity and subcellular localization of Src kinases.Mol Biol Cell. 1999 Oct;10(10):3489-505. doi: 10.1091/mbc.10.10.3489. Mol Biol Cell. 1999. PMID: 10512882 Free PMC article.
-
Signal Transduction Mechanisms of Focal Adhesions: Src and FAK-Mediated Cell Response.Front Biosci (Landmark Ed). 2024 Nov 20;29(11):392. doi: 10.31083/j.fbl2911392. Front Biosci (Landmark Ed). 2024. PMID: 39614431 Review.
-
Signal transduction by focal adhesion kinase in cancer.Cancer Metastasis Rev. 2009 Jun;28(1-2):35-49. doi: 10.1007/s10555-008-9165-4. Cancer Metastasis Rev. 2009. PMID: 19169797 Review.
Cited by
-
pHocal adhesion kinase regulation is on a FERM foundation.J Cell Biol. 2013 Sep 16;202(6):833-6. doi: 10.1083/jcb.201308034. J Cell Biol. 2013. PMID: 24043698 Free PMC article.
-
Conformational dynamics of the focal adhesion targeting domain control specific functions of focal adhesion kinase in cells.J Biol Chem. 2015 Jan 2;290(1):478-91. doi: 10.1074/jbc.M114.593632. Epub 2014 Nov 12. J Biol Chem. 2015. PMID: 25391654 Free PMC article.
-
Focal adhesion kinase signaling in unexpected places.Curr Opin Cell Biol. 2017 Apr;45:24-30. doi: 10.1016/j.ceb.2017.01.003. Epub 2017 Feb 16. Curr Opin Cell Biol. 2017. PMID: 28213315 Free PMC article. Review.
References
-
- Schlaepfer DD, Hunter T. Focal Adhesion Kinase Overexpression Enhances Ras-dependent Integrin Signaling to ERK2/Mitogen-activated Protein Kinase through Interactions with and Activation of c-Src. The Journal of Biological Chemistry. 1997;272:13189–13195. - PubMed
-
- Ren X, Kiosses W, Sieg D, Otey C, Schlaepfer D, Schwartz M. Focal adhesion kinase suppresses Rho activity to promote focal adhesion turnover. J Cell Sci. 2000;113:3673–3678. - PubMed
-
- Ilic D, Furuta Y, Kanazawa S, Takeda N, Sobue K, Nakatsuji N, Nomura S, Fujimoto J, Okada M, Yamamoto T, Aizawa S. Reduced cell motility and enhanced focal adhesion contact formation in cells from FAK-deficient mice. Nature. 1995;377:539–544. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

