Oligonucleotide-templated reactions for sensing nucleic acids
- PMID: 22374329
- PMCID: PMC6268776
- DOI: 10.3390/molecules17032446
Oligonucleotide-templated reactions for sensing nucleic acids
Abstract
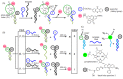
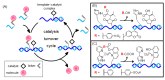
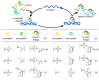
Oligonucleotide-templated reactions are useful for applying nucleic acid sensing. Various chemistries for oligonucleotide-templated reaction have been reported so far. Major scientific interests are focused on the development of signal amplification systems and signal generation systems. We introduce the recent advances of oligonucleotide-templated reaction in consideration of the above two points.
Figures
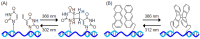
Similar articles
-
Oligonucleotide-templated reactions based on Peptide Nucleic Acid (PNA) probes: concept and biomedical applications.Bioorg Med Chem. 2014 Aug 15;22(16):4395-8. doi: 10.1016/j.bmc.2014.05.071. Epub 2014 Jun 12. Bioorg Med Chem. 2014. PMID: 24957880
-
Nucleic acid templated reactions: consequences of probe reactivity and readout strategy for amplified signaling and sequence selectivity.Chemistry. 2009 Jul 6;15(27):6723-30. doi: 10.1002/chem.200900025. Chemistry. 2009. PMID: 19496097
-
Accelerating Turnover Frequency in Nucleic Acid Templated Reactions.Bioconjug Chem. 2018 Jan 17;29(1):158-163. doi: 10.1021/acs.bioconjchem.7b00663. Epub 2017 Dec 12. Bioconjug Chem. 2018. PMID: 29178795
-
ECHO probes: a concept of fluorescence control for practical nucleic acid sensing.Chem Soc Rev. 2011 Dec;40(12):5815-28. doi: 10.1039/c1cs15025a. Epub 2011 Jun 10. Chem Soc Rev. 2011. PMID: 21660343 Review.
-
Amplification by nucleic acid-templated reactions.Org Biomol Chem. 2014 May 14;12(18):2821-33. doi: 10.1039/c4ob00096j. Org Biomol Chem. 2014. PMID: 24671414 Review.
Cited by
-
Chemical ligation of oligonucleotides using an electrophilic phosphorothioester.Nucleic Acids Res. 2017 Jul 7;45(12):7042-7048. doi: 10.1093/nar/gkx459. Nucleic Acids Res. 2017. PMID: 28520986 Free PMC article.
-
Combining recombinase polymerase amplification and DNA-templated reaction for SARS-CoV-2 sensing with dual fluorescence and lateral flow assay output.Biopolymers. 2022 Apr;113(4):e23485. doi: 10.1002/bip.23485. Epub 2022 Jan 13. Biopolymers. 2022. PMID: 35023571 Free PMC article.
-
Kinase-templated abiotic reaction.Chem Sci. 2017 Jul 1;8(7):5119-5125. doi: 10.1039/c7sc01416c. Epub 2017 May 24. Chem Sci. 2017. PMID: 28970898 Free PMC article.
-
DNA-Dye-Conjugates: Conformations and Spectra of Fluorescence Probes.PLoS One. 2016 Jul 28;11(7):e0160229. doi: 10.1371/journal.pone.0160229. eCollection 2016. PLoS One. 2016. PMID: 27467071 Free PMC article.
-
Rapid oligonucleotide-templated fluorogenic tetrazine ligations.Nucleic Acids Res. 2013 Aug;41(15):e148. doi: 10.1093/nar/gkt540. Epub 2013 Jun 17. Nucleic Acids Res. 2013. PMID: 23775794 Free PMC article.
References
-
- Lewis R.J., Hanawalt P.C. Ligation of oligonucleotides by pyrimidine dimers—A missing “link” in the origin of life? Nature. 1982;298:393–396. - PubMed
-
- Churchill M.E., Peak J.G., Peak M.J. Correlation between cell survival and DNA single-strand break repair proficiency in the Chinese hamster ovary cell lines AA8 and EM9 irradiated with 365-nm ultraviolet-A radiation. Photochem. Photobiol. 1991;53:229–236. doi: 10.1111/j.1751-1097.1991.tb03927.x. - DOI - PubMed
-
- Albagli D., van Atta R., Cheng P., Huan B.F., Wood M.L. Chemical amplification (CHAMP) by a continuous, self-replicating oligonucleotide-based system. J. Am. Chem. Soc. 1999;121:6954–6955. doi: 10.1021/ja990434y. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

