The brain matters: effects of descending signals on motor control
- PMID: 22378172
- PMCID: PMC3362288
- DOI: 10.1152/jn.00107.2012
The brain matters: effects of descending signals on motor control
Abstract
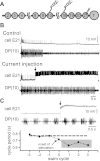
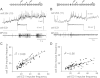
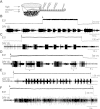
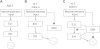
The ability of nerve cords and spinal cords to exhibit fictive rhythmic locomotion in the absence of the brain is well-documented in numerous species. Although the brain is important for modulating the fictive motor output, it is broadly assumed that the functional properties of neuronal circuits identified in simplified preparations are conserved with the brain attached. We tested this assumption by examining the properties of a novel interneuron recently identified in the leech (Hirudo verbana) nerve cord. This neuron, cell E21, initiates and drives stereotyped fictive swimming activity in preparations of the isolated leech nerve cord deprived of the head brain. We report that, contrary to expectation, the motor output generated when cell E21 is stimulated in preparations with the brain attached is highly variable. Swim frequency and episode duration are increased in some of these preparations and decreased in others. Cell E21 controls swimming, in part, via excitatory synaptic interactions with cells 204, previously identified gating neurons that reliably initiate and strongly enhance leech swimming activity when the brain is absent. We found that in preparations with the brain present, the magnitude of the synaptic interaction from cell E21 to cell 204 is reduced by 50% and that cell 204-evoked responses also were highly variable. Intriguingly, most of this variability disappeared in semi-intact preparations. We conclude that neuronal circuit properties identified in reduced preparations might be fundamentally altered from those that occur in more physiological conditions.
Figures





References
-
- Arshavsky YI, Deliagina TG, Orlovsky GN. The swimming circuit in the pteropod mollusc Clione limacina. In: Handbook of Brain Microcircuits, edited by Shepherd GM, Grillner S. New York: Oxford Univ. Press, 2010, p. 474–479
-
- Böhm H, Schildberger K. Brain neurones involved in the control of walking in the cricket Gryllus bimaculatus. J Exp Biol 166: 113–130, 1992
-
- Briggman KL, Abarbanel HD, Kristan WB., Jr Optical images of neuronal populations during decision making. Science 304: 896–901, 2005 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

