Modeling the 5-fluorouracil area under the curve versus dose relationship to develop a pharmacokinetic dosing algorithm for colorectal cancer patients receiving FOLFOX6
- PMID: 22382460
- PMCID: PMC3316912
- DOI: 10.1634/theoncologist.2011-0357
Modeling the 5-fluorouracil area under the curve versus dose relationship to develop a pharmacokinetic dosing algorithm for colorectal cancer patients receiving FOLFOX6
Abstract
Background: 5-Fluorouracil (5-FU) is administered based on standard body surface area (BSA) dosing. BSA administration results in highly variable exposure, measured as the area under the concentration-time curve (AUC). An immunoassay (OnDose®; Myriad Genetic Laboratories, Inc., Salt Lake City, UT) that measures plasma 5-FU concentration and reports an AUC in mg · h/L has been developed to optimize therapy using pharmacokinetic (PK) dosing. The results of an analysis to model the 5-FU AUC-dose relationship are presented.
Methods: A set of 589 sequential patients from a clinical database receiving 5-FU, leucovorin, and oxaliplatin (the FOLFOX6 regimen) for colorectal cancer (CRC) treatment was analyzed. A subset including only patients who had at least two consecutive cycles tested, received 1,600-3,600 mg/m2 of continuous infusion 5-FU during the initial test cycle, and had a blood sample collected after ≥18 hours, was used to conduct regression modeling of the change in AUC versus change in dose.
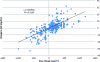
Results: A simple regression model with R(2) = 0.51 developed over n = 307 cycle-pair observations characterizes the AUC-Dose relationship as: change in AUC = 0.02063 * dose change. The model suggests that dose changes in the range of 145-727 mg/m2 would be sufficient to adjust the AUC to a potential therapeutic threshold of >20 mg · h/L for most patients.
Conclusions: 5-FU is an ideal candidate for PK dose optimization. Because individual factors other than dose change may also affect the change in AUC, longitudinal PK monitoring in all cycles and dose adjustment to ensure AUC in the desired range of 20-30 mg · h/L are recommended.
Conflict of interest statement
Figures
Similar articles
-
Personalized dosing via pharmacokinetic monitoring of 5-fluorouracil might reduce toxicity in early- or late-stage colorectal cancer patients treated with infusional 5-fluorouracil-based chemotherapy regimens.Clin Colorectal Cancer. 2014 Jun;13(2):119-26. doi: 10.1016/j.clcc.2013.11.001. Epub 2013 Nov 20. Clin Colorectal Cancer. 2014. PMID: 24461492
-
Individual fluorouracil dose adjustment in FOLFOX based on pharmacokinetic follow-up compared with conventional body-area-surface dosing: a phase II, proof-of-concept study.Clin Colorectal Cancer. 2012 Dec;11(4):263-7. doi: 10.1016/j.clcc.2012.05.004. Epub 2012 Jun 9. Clin Colorectal Cancer. 2012. PMID: 22683364 Clinical Trial.
-
Preliminary observations indicate variable patterns of plasma 5-fluorouracil (5-FU) levels during dose optimization of infusional 5-FU in colorectal cancer patients.Cancer Biol Ther. 2011 Oct 1;12(7):557-68. doi: 10.4161/cbt.12.7.18059. Epub 2011 Oct 1. Cancer Biol Ther. 2011. PMID: 21931273 Clinical Trial.
-
The evolving role of oxaliplatin in the management of colorectal cancer.Colorectal Dis. 2003 Nov;5 Suppl 3:10-9. doi: 10.1046/j.1463-1318.5.s3.3.x. Colorectal Dis. 2003. PMID: 23573556 Review.
-
Phenotype versus genotype to optimize cancer dosing in the clinical setting-focus on 5-fluorouracil and tyrosine kinase inhibitors.Pharmacol Res Perspect. 2024 Apr;12(2):e1182. doi: 10.1002/prp2.1182. Pharmacol Res Perspect. 2024. PMID: 38429945 Free PMC article. Review.
Cited by
-
Pharmacokinetic and Pharmacodynamic Analyses of 5-Fluorouracil in East-Asian Patients with Nasopharyngeal Carcinoma.Clin Pharmacokinet. 2016 Oct;55(10):1205-1216. doi: 10.1007/s40262-016-0395-2. Clin Pharmacokinet. 2016. PMID: 27138786 Clinical Trial.
-
Therapeutic drug monitoring for cytotoxic anticancer drugs: Principles and evidence-based practices.Front Oncol. 2022 Dec 8;12:1015200. doi: 10.3389/fonc.2022.1015200. eCollection 2022. Front Oncol. 2022. PMID: 36568145 Free PMC article. Review.
-
Implementation of pharmacogenetic testing in oncology: DPYD-guided dosing to prevent fluoropyrimidine toxicity in British Columbia.Front Pharmacol. 2023 Sep 8;14:1257745. doi: 10.3389/fphar.2023.1257745. eCollection 2023. Front Pharmacol. 2023. PMID: 37745065 Free PMC article.
-
Drug-Drug Interactions of Irinotecan, 5-Fluorouracil, Folinic Acid and Oxaliplatin and Its Activity in Colorectal Carcinoma Treatment.Molecules. 2020 Jun 4;25(11):2614. doi: 10.3390/molecules25112614. Molecules. 2020. PMID: 32512790 Free PMC article.
-
Prognostic value of the micronucleus assay for clinical endpoints in neoadjuvant radiochemotherapy for rectal cancer.BMC Cancer. 2021 Mar 4;21(1):219. doi: 10.1186/s12885-021-07914-5. BMC Cancer. 2021. PMID: 33663399 Free PMC article.
References
-
- Evans WE, Relling MV. Clinical pharmacokinetics-pharmacodynamics of anticancer drugs. Clin Pharmacokinet. 1989;16:327–336. - PubMed
-
- Masson E, Zamboni WC. Pharmacokinetic optimisation of cancer chemotherapy. Effect on outcomes. Clin Pharmacokinet. 1997;32:324–343. - PubMed
-
- Galpin AJ, Evans WE. Therapeutic drug monitoring in cancer management. Clin Chem. 1993;39:2419–2430. - PubMed
-
- Lamya Alnaim. Individualization of 5-Fluorouracil in the Treatment of Colorectal Cancer. SRX Pharmacology. vol. 2010, Article ID 352491, 12 pages, 2010. doi: 10.3814/2010/352491. - DOI
-
- Ploylearmsaeng SA, Fuhr U, Jetter A. How may anticancer chemotherapy with fluorouracil be individualised. Clin Pharmacokinet. 2006;45:567–592. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical