The ebola virus interferon antagonist VP24 directly binds STAT1 and has a novel, pyramidal fold
- PMID: 22383882
- PMCID: PMC3285596
- DOI: 10.1371/journal.ppat.1002550
The ebola virus interferon antagonist VP24 directly binds STAT1 and has a novel, pyramidal fold
Erratum in
- PLoS Pathog. 2013 Dec;9(12). doi:10.1371/annotation/360ddc68-0313-4eae-af24-043cc040c52d
Abstract
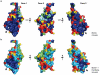
Ebolaviruses cause hemorrhagic fever with up to 90% lethality and in fatal cases, are characterized by early suppression of the host innate immune system. One of the proteins likely responsible for this effect is VP24. VP24 is known to antagonize interferon signaling by binding host karyopherin α proteins, thereby preventing them from transporting the tyrosine-phosphorylated transcription factor STAT1 to the nucleus. Here, we report that VP24 binds STAT1 directly, suggesting that VP24 can suppress at least two distinct branches of the interferon pathway. Here, we also report the first crystal structures of VP24, derived from different species of ebolavirus that are pathogenic (Sudan) and nonpathogenic to humans (Reston). These structures reveal that VP24 has a novel, pyramidal fold. A site on a particular face of the pyramid exhibits reduced solvent exchange when in complex with STAT1. This site is above two highly conserved pockets in VP24 that contain key residues previously implicated in virulence. These crystal structures and accompanying biochemical analysis map differences between pathogenic and nonpathogenic viruses, offer templates for drug design, and provide the three-dimensional framework necessary for biological dissection of the many functions of VP24 in the virus life cycle.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
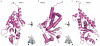
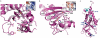
References
-
- Retuya TJA, Miranda MEG, Ksiazek TG, Khan AS, Sanchez A, et al. Is the ebola-reston virus a threat to occupationally exposed humans? J Clin Epidemiol. 1997;50:32S.
-
- Sanchez A, Khan AS, Zaki SR, Nabel GJ, Ksiazek TG, et al. Filoviridae: Marburg and Ebola Viruses. In: Fields BN, Knipe DM, Howley PM, Griffin DE, Lamb RA, et al., editors. Fields virology. 4 ed. Philadelphia: Lippincott Williams & Wilkins; 2001. pp. 1279–1304.
-
- Baize S, Leroy EM, Georges-Courbot MC, Capron M, Lansoud-Soukate J, et al. Defective humoral responses and extensive intravascular apoptosis are associated with fatal outcome in Ebola virus-infected patients. Nat Med. 1999;5:423–426. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- NS070899/NS/NINDS NIH HHS/United States
- AI081982/AI/NIAID NIH HHS/United States
- R01 AI081982/AI/NIAID NIH HHS/United States
- R01 GM020501/GM/NIGMS NIH HHS/United States
- S10 RR029388/RR/NCRR NIH HHS/United States
- R43 AI1088843/AI/NIAID NIH HHS/United States
- 5T32 NS041219/NS/NINDS NIH HHS/United States
- GM020501/GM/NIGMS NIH HHS/United States
- T32 AI007244/AI/NIAID NIH HHS/United States
- R01 NS070899/NS/NINDS NIH HHS/United States
- GM093325/GM/NIGMS NIH HHS/United States
- R01 GM093325/GM/NIGMS NIH HHS/United States
- F32 GM020501/GM/NIGMS NIH HHS/United States
- GM066170/GM/NIGMS NIH HHS/United States
- AI2008031/AI/NIAID NIH HHS/United States
- RR029388/RR/NCRR NIH HHS/United States
- T32 NS041219/NS/NINDS NIH HHS/United States
- R01 GM066170/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

