Characterization of monomeric intermediates during VSV glycoprotein structural transition
- PMID: 22383886
- PMCID: PMC3285605
- DOI: 10.1371/journal.ppat.1002556
Characterization of monomeric intermediates during VSV glycoprotein structural transition
Abstract
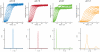
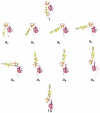
Entry of enveloped viruses requires fusion of viral and cellular membranes, driven by conformational changes of viral glycoproteins. Crystal structures provide static pictures of pre- and post-fusion conformations of these proteins but the transition pathway remains elusive. Here, using several biophysical techniques, including analytical ultracentrifugation, circular dichroïsm, electron microscopy and small angle X-ray scattering, we have characterized the low-pH-induced fusogenic structural transition of a soluble form of vesicular stomatitis virus (VSV) glycoprotein G ectodomain (G(th), aa residues 1-422, the fragment that was previously crystallized). While the post-fusion trimer is the major species detected at low pH, the pre-fusion trimer is not detected in solution. Rather, at high pH, G(th) is a flexible monomer that explores a large conformational space. The monomeric population exhibits a marked pH-dependence and adopts more elongated conformations when pH decreases. Furthermore, large relative movements of domains are detected in absence of significant secondary structure modification. Solution studies are complemented by electron micrographs of negatively stained viral particles in which monomeric ectodomains of G are observed at the viral surface at both pH 7.5 and pH 6.7. We propose that the monomers are intermediates during the conformational change and thus that VSV G trimers dissociate at the viral surface during the structural transition.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

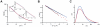



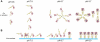
References
-
- Bullough PA, Hughson FM, Skehel JJ, Wiley DC. Structure of influenza haemagglutinin at the pH of membrane fusion. Nature. 1994;371:37–43. - PubMed
-
- Skehel JJ, Wiley DC. Coiled coils in both intracellular vesicle and viral membrane fusion. Cell. 1998;95:871–874. - PubMed
-
- Rey FA, Heinz FX, Mandl C, Kunz C, Harrison SC. The envelope glycoprotein from tick-borne encephalitis virus at 2 A resolution. Nature. 1995;375:291–298. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

