Msx homeobox genes critically regulate embryo implantation by controlling paracrine signaling between uterine stroma and epithelium
- PMID: 22383889
- PMCID: PMC3285581
- DOI: 10.1371/journal.pgen.1002500
Msx homeobox genes critically regulate embryo implantation by controlling paracrine signaling between uterine stroma and epithelium
Abstract
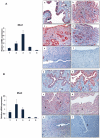
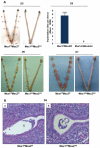
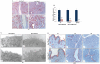
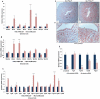
The mammalian Msx homeobox genes, Msx1 and Msx2, encode transcription factors that control organogenesis and tissue interactions during embryonic development. We observed overlapping expression of these factors in uterine epithelial and stromal compartments of pregnant mice prior to embryo implantation. Conditional ablation of both Msx1 and Msx2 in the uterus resulted in female infertility due to a failure in implantation. In these mutant mice (Msx1/2(d/d)), the uterine epithelium exhibited persistent proliferative activity and failed to attach to the embryos. Gene expression profiling of uterine epithelium and stroma of Msx1/2(d/d) mice revealed an elevated expression of several members of the Wnt gene family in the preimplantation uterus. Increased canonical Wnt signaling in the stromal cells activated β-catenin, stimulating the production of a subset of fibroblast growth factors (FGFs) in these cells. The secreted FGFs acted in a paracrine manner via the FGF receptors in the epithelium to promote epithelial proliferation, thereby preventing differentiation of this tissue and creating a non-receptive uterus refractory to implantation. Collectively, these findings delineate a unique signaling network, involving Msx1/2, Wnts, and FGFs, which operate in the uterus at the time of implantation to control the mesenchymal-epithelial dialogue critical for successful establishment of pregnancy.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Carson DD, Bagchi I, Dey SK, Enders AC, Fazleabas AT, et al. Embryo Implantation. Developmental Biology. 2000;223:217–237. - PubMed
-
- Dey SK, Lim H, Das SK, Reese J, Paria BC, et al. Molecular Cues to Implantation. Endocr Rev. 2004;25:341–373. - PubMed
-
- Rubel CA, Jeong J-W, Tsai SY, Lydon JP, DeMayo FJ. Epithelial-Stromal Interaction and Progesterone Receptors in the Mouse Uterus. Semin Reprod Med. 2010;28:027–035. - PubMed
-
- Lee KY, Jeong J-W, Tsai SY, Lydon JP, DeMayo FJ. Mouse models of implantation. Trends in Endocrinology & Metabolism. 2007;18:234–239. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

