Wnt5a is strongly expressed at the leading edge in non-melanoma skin cancer, forming active gradients, while canonical Wnt signalling is repressed
- PMID: 22384081
- PMCID: PMC3285195
- DOI: 10.1371/journal.pone.0031827
Wnt5a is strongly expressed at the leading edge in non-melanoma skin cancer, forming active gradients, while canonical Wnt signalling is repressed
Abstract
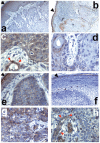
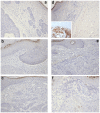
Wnt5a is one of the so-called non-canonical Wnt ligands which do not act through β-catenin. In normal development, Wnt5a is secreted and directs the migration of target cells along concentration gradients. The effect of Wnt5a on target cells is regulated by many factors, including the expression level of inhibitors and receptors. Dysregulated Wnt5a signalling facilitates invasion of multiple tumor types into adjacent tissue. However, the expression and distribution of Wnt5a in cutaneous squamous cell carcinoma (SCC) and basal cell carcinoma (BCC), as well as the effect of Wnt5a on keratinocyte migration has not been studied in detail to date. We here report that Wnt5a is upregulated in SCC and BCC and localised to the leading edge of tumors, as well as tumor-associated fibroblasts. The Wnt5a-triggered bundling of its receptor Fzd3 provides evidence of Wnt5a concentration gradients projecting into the tumor. In vitro migration assays show that Wnt5a concentration gradients determine its effect on keratinoctye migration: While chemotactic migration is inhibited by Wnt5a present in homogenous concentrations, it is enhanced in the presence of a Wnt5a gradient. Expression profiling of the Wnt pathway shows that the upregulation of Wnt5a in SCC is coupled to repression of canonical Wnt signalling. This is confirmed by immunohistochemistry showing lack of nuclear β-catenin, as well as absent accumulation of Axin2. Since both types of Wnt signalling act mutually antogonistically at multiple levels, the concurrent repression of canonical Wnt signalling suggests hyper-active Wnt5a signal transduction. Significantly, this combination of gene dysregulation is not observed in the benign hyperproliferative inflammatory skin disease psoriasis. Collectively, our data strongly suggest that Wnt5a signalling contributes to tissue invasion by non-melanoma skin cancer.
Conflict of interest statement
Figures







Similar articles
-
Canonical Wnt signaling is antagonized by noncanonical Wnt5a in hepatocellular carcinoma cells.Mol Cancer. 2009 Oct 22;8:90. doi: 10.1186/1476-4598-8-90. Mol Cancer. 2009. PMID: 19849855 Free PMC article.
-
Migration and invasion of oral squamous carcinoma cells is promoted by WNT5A, a regulator of cancer progression.J Oral Pathol Med. 2015 Nov;44(10):776-84. doi: 10.1111/jop.12292. Epub 2014 Dec 2. J Oral Pathol Med. 2015. PMID: 25459554
-
Evidence for altered Wnt signaling in psoriatic skin.J Invest Dermatol. 2010 Jul;130(7):1849-59. doi: 10.1038/jid.2010.67. Epub 2010 Apr 8. J Invest Dermatol. 2010. PMID: 20376066 Free PMC article.
-
Wnt5a: its signalling, functions and implication in diseases.Acta Physiol (Oxf). 2012 Jan;204(1):17-33. doi: 10.1111/j.1748-1716.2011.02294.x. Epub 2011 Apr 22. Acta Physiol (Oxf). 2012. PMID: 21518267 Review.
-
Hear the Wnt Ror: how melanoma cells adjust to changes in Wnt.Pigment Cell Melanoma Res. 2009 Dec;22(6):724-39. doi: 10.1111/j.1755-148X.2009.00627.x. Epub 2009 Aug 25. Pigment Cell Melanoma Res. 2009. PMID: 19708915 Free PMC article. Review.
Cited by
-
Mesenchymal-transitioned cancer cells instigate the invasion of epithelial cancer cells through secretion of WNT3 and WNT5B.Cancer Sci. 2014 Mar;105(3):281-9. doi: 10.1111/cas.12336. Epub 2014 Jan 29. Cancer Sci. 2014. PMID: 24344732 Free PMC article.
-
Mathematical Modelling of Molecular Pathways Enabling Tumour Cell Invasion and Migration.PLoS Comput Biol. 2015 Nov 3;11(11):e1004571. doi: 10.1371/journal.pcbi.1004571. eCollection 2015 Nov. PLoS Comput Biol. 2015. PMID: 26528548 Free PMC article.
-
Epigenetic loss of putative tumor suppressor SFRP3 correlates with poor prognosis of lung adenocarcinoma patients.Epigenetics. 2018;13(3):214-227. doi: 10.1080/15592294.2016.1229730. Epub 2018 Apr 18. Epigenetics. 2018. PMID: 27623992 Free PMC article.
-
Wnt Signaling Pathways in Keratinocyte Carcinomas.Cancers (Basel). 2019 Aug 21;11(9):1216. doi: 10.3390/cancers11091216. Cancers (Basel). 2019. PMID: 31438551 Free PMC article. Review.
-
Functionally distinct cancer-associated fibroblast subpopulations establish a tumor promoting environment in squamous cell carcinoma.Nat Commun. 2023 Sep 5;14(1):5413. doi: 10.1038/s41467-023-41141-9. Nat Commun. 2023. PMID: 37669956 Free PMC article.
References
-
- van Amerongen R, Nusse R. Towards an integrated view of Wnt signaling in development. Development. 2009;136:3205–3214. - PubMed
-
- Verkaar F, Zaman GJ. A model for signaling specificity of Wnt/Frizzled combinations through co-receptor recruitment. FEBS Lett. 2010;584:3850–3854. - PubMed
-
- Nishita M, Enomoto M, Yamagata K, Minami Y. Cell/tissue-tropic functions of Wnt5a signaling in normal and cancer cells. Trends Cell Biol. 2010;20:346–354. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

