In vivo fluorescence lifetime imaging monitors binding of specific probes to cancer biomarkers
- PMID: 22384092
- PMCID: PMC3285647
- DOI: 10.1371/journal.pone.0031881
In vivo fluorescence lifetime imaging monitors binding of specific probes to cancer biomarkers
Abstract
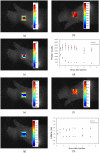
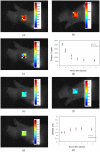
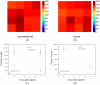
One of the most important factors in choosing a treatment strategy for cancer is characterization of biomarkers in cancer cells. Particularly, recent advances in Monoclonal Antibodies (MAB) as primary-specific drugs targeting tumor receptors show that their efficacy depends strongly on characterization of tumor biomarkers. Assessment of their status in individual patients would facilitate selection of an optimal treatment strategy, and the continuous monitoring of those biomarkers and their binding process to the therapy would provide a means for early evaluation of the efficacy of therapeutic intervention. In this study we have demonstrated for the first time in live animals that the fluorescence lifetime can be used to detect the binding of targeted optical probes to the extracellular receptors on tumor cells in vivo. The rationale was that fluorescence lifetime of a specific probe is sensitive to local environment and/or affinity to other molecules. We attached Near-InfraRed (NIR) fluorescent probes to Human Epidermal Growth Factor 2 (HER2/neu)-specific Affibody molecules and used our time-resolved optical system to compare the fluorescence lifetime of the optical probes that were bound and unbound to tumor cells in live mice. Our results show that the fluorescence lifetime changes in our model system delineate HER2 receptor bound from the unbound probe in vivo. Thus, this method is useful as a specific marker of the receptor binding process, which can open a new paradigm in the "image and treat" concept, especially for early evaluation of the efficacy of the therapy.
Conflict of interest statement
Figures

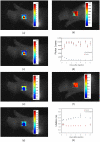
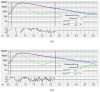
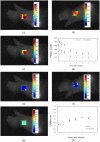
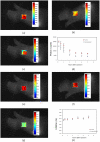



Similar articles
-
In vivo assessment of HER2 receptor density in HER2-positive tumors by near-infrared imaging, using repeated injections of the fluorescent probe.Technol Cancer Res Treat. 2014 Oct;13(5):427-34. doi: 10.7785/tcrtexpress.2013.600265. Epub 2013 Aug 31. Technol Cancer Res Treat. 2014. PMID: 24000992 Free PMC article.
-
Using in vivo fluorescence lifetime imaging to detect HER2-positive tumors.EJNMMI Res. 2018 Apr 4;8(1):26. doi: 10.1186/s13550-018-0384-6. EJNMMI Res. 2018. PMID: 29619584 Free PMC article.
-
Affibody-DyLight conjugates for in vivo assessment of HER2 expression by near-infrared optical imaging.PLoS One. 2012;7(7):e41016. doi: 10.1371/journal.pone.0041016. Epub 2012 Jul 20. PLoS One. 2012. PMID: 22911732 Free PMC article.
-
Dual-labeled trastuzumab-based imaging agent for the detection of human epidermal growth factor receptor 2 overexpression in breast cancer.J Nucl Med. 2007 Sep;48(9):1501-10. doi: 10.2967/jnumed.107.042234. J Nucl Med. 2007. PMID: 17785729
-
Using in-vivo fluorescence imaging in personalized cancer diagnostics and therapy, an image and treat paradigm.Technol Cancer Res Treat. 2011 Dec;10(6):549-60. doi: 10.1177/153303461101000605. Technol Cancer Res Treat. 2011. PMID: 22066595 Free PMC article. Review.
Cited by
-
Multiscale BerEp4 molecular imaging of microtumor phantoms: toward theranostics for basal cell carcinoma.Mol Imaging. 2014;13:10.2310/7290.2014.00016. doi: 10.2310/7290.2014.00016. Mol Imaging. 2014. PMID: 25022347 Free PMC article.
-
Molecular probes for fluorescence lifetime imaging.Bioconjug Chem. 2015 Jun 17;26(6):963-74. doi: 10.1021/acs.bioconjchem.5b00167. Epub 2015 May 22. Bioconjug Chem. 2015. PMID: 25961514 Free PMC article. Review.
-
Peptide-fluorescent bacteria complex as luminescent reagents for cancer diagnosis.PLoS One. 2013;8(1):e54467. doi: 10.1371/journal.pone.0054467. Epub 2013 Jan 18. PLoS One. 2013. PMID: 23349898 Free PMC article.
-
Phasor Analysis of Fluorescence Lifetime Enables Quantitative Multiplexed Molecular Imaging of Three Probes.Anal Chem. 2022 Oct 18;94(41):14185-14194. doi: 10.1021/acs.analchem.2c02149. Epub 2022 Oct 3. Anal Chem. 2022. PMID: 36190014 Free PMC article.
-
In vivo assessment of HER2 receptor density in HER2-positive tumors by near-infrared imaging, using repeated injections of the fluorescent probe.Technol Cancer Res Treat. 2014 Oct;13(5):427-34. doi: 10.7785/tcrtexpress.2013.600265. Epub 2013 Aug 31. Technol Cancer Res Treat. 2014. PMID: 24000992 Free PMC article.
References
-
- Allison M. The HER2 testing conundrum. Nature Biotechnology. 2010;28:117–119. - PubMed
-
- Nutt R, Vento LJ, Ridinger MH. In vivo molecular imaging biomarkers: clinical pharmacology's new “PET”? Clin Pharmacol Ther. 2007;81:792–795. - PubMed
-
- Bremer C, Ntziachristos V, Weissleder R. Optical-based molecular imaging: contrast agents and potential medical applications. Eur Radiol. 2003;13:231–243. - PubMed
-
- Corlu A, Choe R, Durduran T, Rosen MA, Schweiger M, et al. Three-dimensional in vivo fluorescence diffuse optical tomography of breast cancer in humans. Opt Express. 2007;15:6696–6716. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

