Pathobiological implications of the expression of EGFR, pAkt, NF-κB and MIC-1 in prostate cancer stem cells and their progenies
- PMID: 22384099
- PMCID: PMC3285632
- DOI: 10.1371/journal.pone.0031919
Pathobiological implications of the expression of EGFR, pAkt, NF-κB and MIC-1 in prostate cancer stem cells and their progenies
Abstract
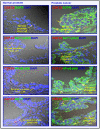
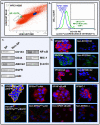
The progression of prostate cancers (PCs) to locally invasive, androgen-independent and metastatic disease states is generally associated with treatment resistance and disease relapse. The present study was undertaken to establish the possibility of using a combination of specific oncogenic products, including epidermal growth factor receptor (EGFR), pAkt, nuclear factor-kappaB (NF-κB) and macrophage inhibitory cytokine-1 (MIC-1) as biomarkers and therapeutic targets for optimizing the management of patients with localized PC at earlier disease stages. The immunohistochemical and immunofluorescence data have revealed that the expression levels of EGFR, Ser(473)-pAkt, NF-κB p65 and MIC-1 proteins were significantly enhanced in the same subset of 76 cases of prostatic adenocarcinoma specimens during the disease progression and these biomarkers were expressed in a small subpopulation of CD133(+) PC cells and the bulk tumor mass of CD133(-) PC cells. Importantly, all of these biomarkers were also overexpressed in 80-100% of 30 PC metastasis bone tissue specimens. Moreover, the results have indicated that the EGF-EGFR signaling pathway can provide critical functions for the self-renewal of side population (SP) cells endowed with stem cell-like features from highly invasive WPE1-NB26 cells. Of therapeutic interest, the targeting of EGFR, pAkt, NF-κB or MIC-1 was also effective at suppressing the basal and EGF-promoted prostasphere formation by SP WPE1-NB26 cells, inducing disintegration of SP cell-derived prostaspheres and decreasing the viability of SP and non-SP WPE1-NB26 cell fractions. Also, the targeting of these oncogenic products induced the caspase-dependent apoptosis in chemoresistant SP WPE1-NB26 cells and enhanced their sensibility to the cytotoxic effects induced by docetaxel. These findings suggest that the combined use of EGFR, pAkt, NF-κB and/or MIC-1 may represent promising strategies for improving the accuracy of current diagnostic and prognostic methods and efficacy of treatments of PC patients in considering the disease heterogeneity, thereby preventing PC progression to metastatic and lethal disease states.
Conflict of interest statement
Figures






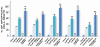
References
-
- Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–1512. - PubMed
-
- Petrylak DP, Tangen CM, Hussain MH, Lara PN, Jr, Jones JA, et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med. 2004;351:1513–1520. - PubMed
-
- Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300. - PubMed
-
- Mimeault M, Batra SK. Recent advances on multiple tumorigenic cascades involved in prostatic cancer progression and targeting therapies. Carcinogenesis. 2006;27:1–22. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

