IP3 3-kinase opposes NGF driven neurite outgrowth
- PMID: 22384237
- PMCID: PMC3284567
- DOI: 10.1371/journal.pone.0032386
IP3 3-kinase opposes NGF driven neurite outgrowth
Abstract
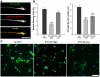
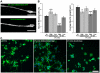
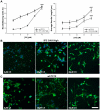
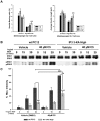
The inositol (1,4,5) trisphosphate 3-kinases comprise a family of enzymes (A, B, and C) that phosphorylate the calcium mobilising molecule inositol (1,4,5) trisphosphate (IP(3)) to generate inositol (1,3,4,5) tetrakisphosphate. This molecule can function as a second messenger, but its roles are not completely understood. The A isoform of inositol (1,4,5) trisphosphate 3-kinase localises to filamentous actin within dendritic spines in the hippocampus and is implicated in the regulation of spine morphology and long term potentiation, however the mechanisms through which it signals in neuronal cells are not completely understood. We have used NGF driven neurite outgrowth from PC12 cells as a platform to examine the impact of signaling via inositol (1,4,5) trisphosphate 3-kinase activity in a neuronal cell. We have found that the catalytic activity of the enzyme opposes neurite outgrowth, whilst pharmacological inhibition of inositol (1,4,5) trisphosphate 3-kinase leads to a significant increase in neurite outgrowth, and we show that the reduction in neurite outgrowth in response to inositol (1,4,5) trisphosphate 3-kinase activity correlates with reduced ERK activity as determined by western blotting using phosphorylation-specific antibodies. Our findings suggest a novel neuronal signaling pathway linking metabolism of IP(3) to signaling via ERK.
Conflict of interest statement
Figures
References
-
- Berridge MJ. Unlocking the secrets of cell signaling. Annu Rev Physiol. 2005;67:1–21. - PubMed
-
- Miller AT, Chamberlain PP, Cooke MP. Beyond IP3: roles for higher order inositol phosphates in immune cell signaling. Cell Cycle. 2008;7:463–467. - PubMed
-
- Pattni K, Banting G. Ins(1,4,5)P3 metabolism and the family of IP3-3Kinases. Cell Signal. 2004;16:643–654. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

