A Genetic Screen for Dominant Enhancers of the Cell-Cycle Regulator α-Endosulfine Identifies Matrimony as a Strong Functional Interactor in Drosophila
- PMID: 22384372
- PMCID: PMC3276179
- DOI: 10.1534/g3.111.001438
A Genetic Screen for Dominant Enhancers of the Cell-Cycle Regulator α-Endosulfine Identifies Matrimony as a Strong Functional Interactor in Drosophila
Abstract
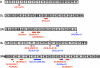
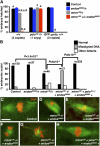
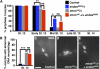
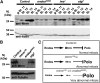
The coordination of cell-cycle events with developmental processes is essential for the reproductive success of organisms. In Drosophila melanogaster, meiosis is tightly coupled to oocyte development, and early embryos undergo specialized S-M mitoses that are supported by maternal products. We previously showed that the small phosphoprotein α-endosulfine (Endos) is required for normal oocyte meiotic maturation and early embryonic mitoses in Drosophila. In this study, we performed a genetic screen for dominant enhancers of endos(00003) and identified several genomic regions that, when deleted, lead to impaired fertility of endos(00003)/+ heterozygous females. We uncovered matrimony (mtrm), which encodes a Polo kinase inhibitor, as a strong dominant enhancer of endos. mtrm(126) +/+ endos(00003) females are sterile because of defects in early embryonic mitoses, and this phenotype is reverted by removal of one copy of polo. These results provide compelling genetic evidence that excessive Polo activity underlies the strong functional interaction between endos(00003) and mtrm(126). Moreover, we show that endos is required for the increased expression of Mtrm in mature oocytes, which is presumably loaded into early embryos. These data are consistent with the model that maternal endos antagonizes Polo function in the early embryo to ensure normal mitoses through its effects on Mtrm expression during late oogenesis. Finally, we also identified genomic deletions that lead to loss of viability of endos(00003)/+ heterozygotes, consistent with recently published studies showing that endos is required zygotically to regulate the cell cycle during development.
Keywords: Drosophila; early embryonic cell cycle; matrimony; polo; α-endosulfine.
Figures

References
-
- Drummond-Barbosa D., Spradling A. C., 2004. Alpha-endosulfine, a potential regulator of insulin secretion, is required for adult tissue growth control in Drosphila. Dev. Biol. 266: 310–321 - PubMed
-
- Gharbi-Ayachi A., Labbe J. C., Burgess A., Vigneron S., Strub J. M., et al. , 2010. The substrate of Greatwall kinase, Arpp19, controls mitosis by inhibiting protein phosphatase 2A. Science 330: 1673–1677 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
