Mechanical stress-activated immune response genes via Sirtuin 1 expression in human periodontal ligament cells
- PMID: 22385246
- PMCID: PMC3390502
- DOI: 10.1111/j.1365-2249.2011.04549.x
Mechanical stress-activated immune response genes via Sirtuin 1 expression in human periodontal ligament cells
Abstract
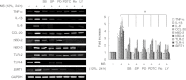
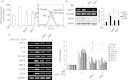
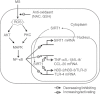
Recently, Sirtuin 1 (SIRT1) has been implicated in the molecular control of ageing and immune response. Although the remodelling of periodontal ligament (PDL) in response to mechanical stress (MS) is mediated by several host factors, including cytokines and chemokines, the transmission of mechanical stimuli into specific cellular activity is still not understood fully. This study aimed to investigate the effects of MS, particularly cyclic strain, on immune response genes, as well as SIRT1 and its signal transduction pathways, in human PDL cells. MS up-regulated the expression of SIRT1 and immune response genes encoding cytokines [tumour necrosis factor (TNF)-α, interleukin (IL)-1β], chemokines [IL-8, monocyte cheoattractant protein (CCL)-20], defensins [human β-defensin (hBD)-2, hBD-3] and Toll-like receptors (TLR-2 and TLR-4) in a force- and time-dependent manner. The SIRT1 inducers resveratrol and isonicotinamide attenuated MS-induced cytokine and chemokine expression, but enhanced the expression of defensins and TLRs. Blockade of SIRT1 activity by the SIRT1 inhibitors sirtinol and nicotinamide and down-regulation of SIRT1 expression by SIRT1 siRNA reduced the stimulatory effects of MS on defensins and TLRs, but increased its effects on cytokines and chemokines. MS induced activation of protein kinase B (Akt), protein kinase C (PKC), nuclear factor (NF)-κB and p38 mitogen-activated protein kinase (MAPK), c-Jun N-terminal kinase (JNK) and extracellular signal-regulated kinase (ERK). Treatment with the anti-oxidants N-acetylcysteine and glutathione inhibited MS-induced reactive oxygen species production and expression of cytokines, chemokines, defensins and TLRs. These results suggest that MS activates human PDL cells to express immune/defence genes encoding cytokines, chemokines, defensins and TLRs via a SIRT1 pathway.
© 2011 The Authors. Clinical and Experimental Immunology © 2011 British Society for Immunology.
Figures





References
-
- Davidovitch Z. Tooth movement. Crit Rev Oral Biol Med. 1991;2:411–50. - PubMed
-
- Tosi MF. Immune responses to infection. J Allergy Clin Immunol. 2005;116:241–9. - PubMed
-
- Charo IF, Ransohoff RM. The many roles of chemokines and chemokine receptors in inflammation. N Engl J Med. 2006;354:610–21. - PubMed
-
- Koyama Y, Mitsui N, Suzuki N, et al. Effect of compressive force on the expression of inflammatory cytokines and their receptors in osteoblastic Saos-2 cells. Arch Oral Biol. 2008;53:488–96. - PubMed
-
- Alhashimi N, Frithiof L, Brudvik P, Bakhiet M. Chemokines are upregulated during orthodontic tooth movement. J Interferon Cytokine Res. 1999;19:1047–52. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

