Order out of disorder: working cycle of an intrinsically unfolded chaperone
- PMID: 22385960
- PMCID: PMC3376891
- DOI: 10.1016/j.cell.2012.01.045
Order out of disorder: working cycle of an intrinsically unfolded chaperone
Abstract
The redox-regulated chaperone Hsp33 protects organisms against oxidative stress that leads to protein unfolding. Activation of Hsp33 is triggered by the oxidative unfolding of its own redox-sensor domain, making Hsp33 a member of a recently discovered class of chaperones that require partial unfolding for full chaperone activity. Here we address the long-standing question of how chaperones recognize client proteins. We show that Hsp33 uses its own intrinsically disordered regions to discriminate between unfolded and partially structured folding intermediates. Binding to secondary structure elements in client proteins stabilizes Hsp33's intrinsically disordered regions, and this stabilization appears to mediate Hsp33's high affinity for structured folding intermediates. Return to nonstress conditions reduces Hsp33's disulfide bonds, which then significantly destabilizes the bound client proteins and in doing so converts them into less-structured, folding-competent client proteins of ATP-dependent foldases. We propose a model in which energy-independent chaperones use internal order-to-disorder transitions to control substrate binding and release.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures
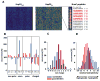
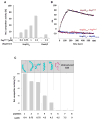
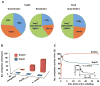
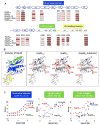


Comment in
-
The unfolding story of a redox chaperone.Cell. 2012 Mar 2;148(5):843-4. doi: 10.1016/j.cell.2012.02.029. Cell. 2012. PMID: 22385952
References
-
- Bowie JU, Sauer RT. Equilibrium dissociation and unfolding of the Arc repressor dimer. Biochemistry. 1989;28:7139–7143. - PubMed
-
- Chen Z, Kurt N, Rajagopalan S, Cavagnero S. Secondary structure mapping of DnaK-bound protein fragments: chain helicity and local helix unwinding at the binding site. Biochemistry. 2006;45:12325–12333. - PubMed
-
- Deuerling E, Schulze-Specking A, Tomoyasu T, Mogk A, Bukau B. Trigger factor and DnaK cooperate in folding of newly synthesized proteins. Nature. 1999;400:693–696. - PubMed
-
- Gaspar AM, Appavou MS, Busch S, Unruh T, Doster W. Dynamics of well-folded and natively disordered proteins in solution: a time-of-flight neutron scattering study. Eur Biophys J. 2008;37:573–582. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

