Neuropeptide delivery to synapses by long-range vesicle circulation and sporadic capture
- PMID: 22385966
- PMCID: PMC3294265
- DOI: 10.1016/j.cell.2011.12.036
Neuropeptide delivery to synapses by long-range vesicle circulation and sporadic capture
Abstract
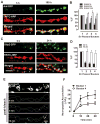
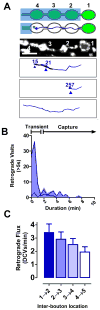
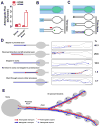
Neurotransmission requires anterograde axonal transport of dense core vesicles (DCVs) containing neuropeptides and active zone components from the soma to nerve terminals. However, it is puzzling how one-way traffic could uniformly supply sequential release sites called en passant boutons. Here, Drosophila neuropeptide-containing DCVs are tracked in vivo for minutes with a new method called simultaneous photobleaching and imaging (SPAIM). Surprisingly, anterograde DCVs typically bypass proximal boutons to accumulate initially in the most distal bouton. Then, excess distal DCVs undergo dynactin-dependent retrograde transport back through proximal boutons into the axon. Just before re-entering the soma, DCVs again reverse for another round of anterograde axonal transport. While circulating over long distances, both anterograde and retrograde DCVs are captured sporadically in en passant boutons. Therefore, vesicle circulation, which includes long-range retrograde transport and inefficient bidirectional capture, overcomes the limitations of one-way anterograde transport to uniformly supply release sites with DCVs.
Copyright © 2012 Elsevier Inc. All rights reserved.
Figures




Comment in
-
Synaptic vesicle distribution by conveyor belt.Cell. 2012 Mar 2;148(5):849-51. doi: 10.1016/j.cell.2012.02.007. Cell. 2012. PMID: 22385955
Similar articles
-
Activity Induces Fmr1-Sensitive Synaptic Capture of Anterograde Circulating Neuropeptide Vesicles.J Neurosci. 2016 Nov 16;36(46):11781-11787. doi: 10.1523/JNEUROSCI.2212-16.2016. J Neurosci. 2016. PMID: 27852784 Free PMC article.
-
Synaptic neuropeptide release by dynamin-dependent partial release from circulating vesicles.Mol Biol Cell. 2015 Jul 1;26(13):2466-74. doi: 10.1091/mbc.E15-01-0002. Epub 2015 Apr 22. Mol Biol Cell. 2015. PMID: 25904335 Free PMC article.
-
Loss of Huntingtin stimulates capture of retrograde dense-core vesicles to increase synaptic neuropeptide stores.Eur J Cell Biol. 2017 Aug;96(5):402-406. doi: 10.1016/j.ejcb.2017.01.001. Epub 2017 Jan 22. Eur J Cell Biol. 2017. PMID: 28129919 Free PMC article.
-
Signaling for vesicle mobilization and synaptic plasticity.Mol Neurobiol. 2008 Feb;37(1):39-43. doi: 10.1007/s12035-008-8014-3. Epub 2008 Apr 30. Mol Neurobiol. 2008. PMID: 18446451 Free PMC article. Review.
-
An anterograde neuroanatomical tracing method that shows the detailed morphology of neurons, their axons and terminals: Immunohistochemical localization of an axonally transported plant lectin, Phaseolus vulgaris-leucoagglutinin (PHA-L).Brain Res. 2016 Aug 15;1645:42-5. doi: 10.1016/j.brainres.2015.12.040. Epub 2016 Jan 12. Brain Res. 2016. PMID: 26790346 Review.
Cited by
-
Differential axonal trafficking of Neuropeptide Y-, LAMP1-, and RAB7-tagged organelles in vivo.Elife. 2022 Dec 2;11:e81721. doi: 10.7554/eLife.81721. Elife. 2022. PMID: 36459486 Free PMC article.
-
Changes in neuropeptide large dense core vesicle trafficking dynamics contribute to adaptive responses to a systemic homeostatic challenge.iScience. 2023 Oct 18;26(11):108243. doi: 10.1016/j.isci.2023.108243. eCollection 2023 Nov 17. iScience. 2023. PMID: 38026155 Free PMC article.
-
In vivo imaging of disease-related mitochondrial dynamics in a vertebrate model system.J Neurosci. 2012 Nov 14;32(46):16203-12. doi: 10.1523/JNEUROSCI.1327-12.2012. J Neurosci. 2012. PMID: 23152604 Free PMC article.
-
Microtubule polarity is instructive for many aspects of neuronal polarity.Dev Biol. 2022 Jun;486:56-70. doi: 10.1016/j.ydbio.2022.03.009. Epub 2022 Mar 25. Dev Biol. 2022. PMID: 35341730 Free PMC article.
-
Live Imaging of Axonal Transport in the Motor Neurons of Drosophila Larvae.Bio Protoc. 2017 Dec 5;7(23):e2631. doi: 10.21769/BioProtoc.2631. eCollection 2017 Dec 5. Bio Protoc. 2017. PMID: 34595299 Free PMC article.
References
-
- Ahmari SE, Buchanan J, Smith SJ. Assembly of presynaptic active zones from cytoplasmic transport packets. Nat Neurosci. 2000;3:445–451. - PubMed
-
- Alonso G, Assenmacher I. Retrograde axoplasmic transport of neurosecretory material. An immunocytochemical and electron-microscopic study of transected axons in normal and colchicine-treated rats. Cell Tissue Res. 1983;233:183–196. - PubMed
-
- Bodmer R, Jan YN. Morphological differentiation of the embryonic peripheral neurons in Drosophila. Roux’s Arch Dev Biol. 1987;196:69–77. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

