Afferent drive elicits ongoing pain in a model of advanced osteoarthritis
- PMID: 22387095
- PMCID: PMC3313555
- DOI: 10.1016/j.pain.2012.01.022
Afferent drive elicits ongoing pain in a model of advanced osteoarthritis
Abstract
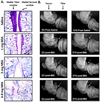
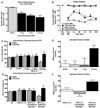
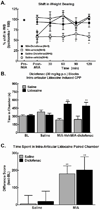
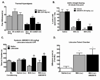
Osteoarthritis (OA) is a chronic condition characterized by pain during joint movement. Additionally, patients with advanced disease experience pain at rest (ie, ongoing pain) that is generally resistant to nonsteroidal antiinflammatory drugs. Injection of monosodium iodoacetate (MIA) into the intraarticular space of the rodent knee is a well-established model of OA that elicits weight-bearing asymmetry and referred tactile and thermal hypersensitivity. Whether ongoing pain is present in this model is unknown. Additionally, the possible relationship of ongoing pain to MIA dose is not known. MIA produced weight asymmetry, joint osteolysis, and cartilage erosion across a range of doses (1, 3, and 4.8 mg). However, only rats treated with the highest dose of MIA showed conditioned place preference to a context paired with intraarticular lidocaine, indicating relief from ongoing pain. Diclofenac blocked the MIA-induced weight asymmetry but failed to block MIA-induced ongoing pain. Systemic AMG9810, a transient receptor potential V1 channel (TRPV1) antagonist, effectively blocked thermal hypersensitivity, but failed to block high-dose MIA-induced weight asymmetry or ongoing pain. Additionally, systemic or intraarticular HC030031, a TRPA1 antagonist, failed to block high-dose MIA-induced weight asymmetry or ongoing pain. Our studies suggest that a high dose of intraarticular MIA induces ongoing pain originating from the site of injury that is dependent on afferent fiber activity but apparently independent of TRPV1 or TRPA1 activation. Identification of mechanisms driving ongoing pain may enable development of improved treatments for patients with severe OA pain and diminish the need for joint replacement surgery.
Copyright © 2012 International Association for the Study of Pain. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
The authors have no conflict of interest to declare.
Figures

Similar articles
-
Effects of Treadmill Exercise on Advanced Osteoarthritis Pain in Rats.Arthritis Rheumatol. 2017 Jul;69(7):1407-1417. doi: 10.1002/art.40101. Epub 2017 Jun 5. Arthritis Rheumatol. 2017. PMID: 28320059 Free PMC article.
-
Ongoing pain in the MIA model of osteoarthritis.Neurosci Lett. 2011 Apr 15;493(3):72-5. doi: 10.1016/j.neulet.2011.01.027. Epub 2011 Jan 15. Neurosci Lett. 2011. PMID: 21241772 Free PMC article.
-
Central Sensitization and Neuropathic Features of Ongoing Pain in a Rat Model of Advanced Osteoarthritis.J Pain. 2016 Mar;17(3):374-82. doi: 10.1016/j.jpain.2015.12.001. Epub 2015 Dec 13. J Pain. 2016. PMID: 26694132 Free PMC article.
-
Differential contributions of peripheral and central mechanisms to pain in a rodent model of osteoarthritis.Sci Rep. 2018 May 8;8(1):7122. doi: 10.1038/s41598-018-25581-8. Sci Rep. 2018. PMID: 29740093 Free PMC article.
-
Augmented pain behavioural responses to intra-articular injection of nerve growth factor in two animal models of osteoarthritis.Ann Rheum Dis. 2014 Sep;73(9):1710-8. doi: 10.1136/annrheumdis-2013-203416. Epub 2013 Jul 13. Ann Rheum Dis. 2014. PMID: 23852764 Free PMC article.
Cited by
-
Pain relief produces negative reinforcement through activation of mesolimbic reward-valuation circuitry.Proc Natl Acad Sci U S A. 2012 Dec 11;109(50):20709-13. doi: 10.1073/pnas.1214605109. Epub 2012 Nov 26. Proc Natl Acad Sci U S A. 2012. PMID: 23184995 Free PMC article.
-
Persistent hyperalgesia in the cisplatin-treated mouse as defined by threshold measures, the conditioned place preference paradigm, and changes in dorsal root ganglia activated transcription factor 3: the effects of gabapentin, ketorolac, and etanercept.Anesth Analg. 2013 Jan;116(1):224-31. doi: 10.1213/ANE.0b013e31826e1007. Epub 2012 Dec 7. Anesth Analg. 2013. PMID: 23223118 Free PMC article.
-
Reward and motivation in pain and pain relief.Nat Neurosci. 2014 Oct;17(10):1304-12. doi: 10.1038/nn.3811. Epub 2014 Sep 25. Nat Neurosci. 2014. PMID: 25254980 Free PMC article. Review.
-
An update on targets for treating osteoarthritis pain: NGF and TRPV1.Curr Treatm Opt Rheumatol. 2020 Sep;6(3):129-145. doi: 10.1007/s40674-020-00146-x. Epub 2020 May 6. Curr Treatm Opt Rheumatol. 2020. PMID: 34178580 Free PMC article.
-
Selective antagonism of TRPA1 produces limited efficacy in models of inflammatory- and neuropathic-induced mechanical hypersensitivity in rats.Mol Pain. 2016 Nov 29;12:1744806916677761. doi: 10.1177/1744806916677761. Print 2016. Mol Pain. 2016. PMID: 27899696 Free PMC article.
References
-
- Argoff CE. Conclusions: chronic pain studies of lidocaine patch 5% using the Neuropathic Pain Scale. Curr Med Res Opin. 2004;20 Suppl 2:S29–S31. - PubMed
-
- Bardo MT, Bevins RA. Conditioned place preference: what does it add to our preclinical understanding of drug reward? Psychopharmacology. 2000;153(1):31–43. - PubMed
-
- Burch F, Codding C, Patel N, Sheldon E. Lidocaine patch 5% improves pain, stiffness, and physical function in osteoarthritis pain patients. A prospective, multicenter, open-label effectiveness trial. Osteoarthritis Cartilage. 2004;12(3):253–255. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

