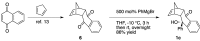Anti-addition mechanism in the intramolecular hydroalkoxylation of alkenes catalyzed by PVP-stabilized nanogold
- PMID: 22388968
- PMCID: PMC6268499
- DOI: 10.3390/molecules17032579
Anti-addition mechanism in the intramolecular hydroalkoxylation of alkenes catalyzed by PVP-stabilized nanogold
Abstract
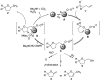
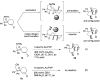
(1R*,4S*,4aR*,9aS*,10S*)-10-Hydroxy-10-phenyl-1,4a,9a,10-tetrahydro-1,4-methanoanthracen-9(4H)-one (1c) was prepared for the elucidation of the reaction mechanism of intramolecular hydroalkoxylation of alkenes catalyzed by gold nanoclusters stabilized by a hydrophilic polymer, poly(N-vinyl-2-pyrrolidone) (Au:PVP). It was found that the reaction proceeded via anti-addition of alcohol to the alkene assisted by p-activation of the gold clusters, which is the same mechanism as the hydroamination by toluenesulfonamides.
Figures
Similar articles
-
Preparation and catalytic activity of poly(N-vinyl-2-pyrrolidone)-protected Au nanoparticles for the aerobic oxidation of glucose.J Nanosci Nanotechnol. 2014 Aug;14(8):5743-51. doi: 10.1166/jnn.2014.8839. J Nanosci Nanotechnol. 2014. PMID: 25935998
-
Aerobic oxidations catalyzed by colloidal nanogold.Chem Asian J. 2011 Mar 1;6(3):736-48. doi: 10.1002/asia.201000611. Epub 2011 Feb 16. Chem Asian J. 2011. PMID: 21328698
-
Magnetically recoverable magnetite/gold catalyst stabilized by poly(N-vinyl-2-pyrrolidone) for aerobic oxidation of alcohols.Molecules. 2010 Dec 29;16(1):149-61. doi: 10.3390/molecules16010149. Molecules. 2010. PMID: 21191318 Free PMC article.
-
Gold and silver catalysis: from organic transformation to bioconjugation.Org Biomol Chem. 2015 Jun 28;13(24):6667-80. doi: 10.1039/c5ob00407a. Org Biomol Chem. 2015. PMID: 25997423 Review.
-
Gold-catalyzed cycloisomerizations of enynes: a mechanistic perspective.Chem Rev. 2008 Aug;108(8):3326-50. doi: 10.1021/cr0684319. Epub 2008 Jul 18. Chem Rev. 2008. PMID: 18636778 Review. No abstract available.
References
-
- Haruta M., Kobayashi T., Sano H., Yamada N. Novel gold catalysts for the oxidation of carbon monoxide at a temperature far below 0 °C. Chem. Lett. 1987:405–408.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources