Subcellular dynamics of multifunctional protein regulation: mechanisms of GAPDH intracellular translocation
- PMID: 22388977
- PMCID: PMC3350569
- DOI: 10.1002/jcb.24113
Subcellular dynamics of multifunctional protein regulation: mechanisms of GAPDH intracellular translocation
Abstract
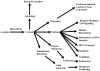
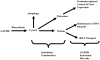
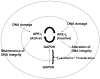
Multidimensional proteins such as glyceraldehyde-3-phosphate dehydrogenase (GAPDH) exhibit distinct activities unrelated to their originally identified functions. Apart from glycolysis, GAPDH participates in iron metabolism, membrane trafficking, histone biosynthesis, the maintenance of DNA integrity and receptor mediated cell signaling. Further, multifunctional proteins exhibit distinct changes in their subcellular localization reflecting their new activities. As such, GAPDH is not only a cytosolic protein but is localized in the membrane, the nucleus, polysomes, the ER and the Golgi. In addition, although the initial subcellular localizations of multifunctional proteins may be of significance, dynamic changes in intracellular distribution may occur as a consequence of those new activities. As such, regulatory mechanisms may exist through which cells control multifunctional protein expression as a function of their subcellular localization. The temporal sequence through which subcellular translocation and the acquisition of new GAPDH functions is considered as well as post-translational modification as a basis for its intracellular transport.
Copyright © 2012 Wiley Periodicals, Inc.
Figures
Similar articles
-
New nuclear functions of the glycolytic protein, glyceraldehyde-3-phosphate dehydrogenase, in mammalian cells.J Cell Biochem. 2005 May 1;95(1):45-52. doi: 10.1002/jcb.20399. J Cell Biochem. 2005. PMID: 15770658 Review.
-
Glyceraldehyde-3-phosphate dehydrogenase interacts with Rab2 and plays an essential role in endoplasmic reticulum to Golgi transport exclusive of its glycolytic activity.J Biol Chem. 2004 Dec 24;279(52):54046-52. doi: 10.1074/jbc.M409472200. Epub 2004 Oct 14. J Biol Chem. 2004. PMID: 15485821
-
Role of glyceraldehyde-3-phosphate dehydrogenase in vesicular transport from golgi apparatus to endoplasmic reticulum.Biochemistry (Mosc). 2008 Jun;73(6):619-25. doi: 10.1134/s0006297908060011. Biochemistry (Mosc). 2008. PMID: 18620527 Review.
-
Subcellular localization of human glyceraldehyde-3-phosphate dehydrogenase is independent of its glycolytic function.Biochim Biophys Acta. 2003 Jun 20;1622(1):50-6. doi: 10.1016/s0304-4165(03)00117-x. Biochim Biophys Acta. 2003. PMID: 12829261
-
The diverse functions of GAPDH: views from different subcellular compartments.Cell Signal. 2011 Feb;23(2):317-23. doi: 10.1016/j.cellsig.2010.08.003. Epub 2010 Aug 19. Cell Signal. 2011. PMID: 20727968 Free PMC article. Review.
Cited by
-
Identification and expression profiling of GAPDH family genes involved in response to Sclerotinia sclerotiorum infection and phytohormones in Brassica napus.Front Plant Sci. 2024 Apr 30;15:1360024. doi: 10.3389/fpls.2024.1360024. eCollection 2024. Front Plant Sci. 2024. PMID: 38745922 Free PMC article.
-
Localization of TFPI-2 in the nucleus modulates MMP-2 gene expression in breast cancer cells.Sci Rep. 2017 Oct 19;7(1):13575. doi: 10.1038/s41598-017-14148-8. Sci Rep. 2017. PMID: 29051606 Free PMC article.
-
Moonlighting Proteins Are Important Players in Cancer Immunology.Front Immunol. 2021 Jan 18;11:613069. doi: 10.3389/fimmu.2020.613069. eCollection 2020. Front Immunol. 2021. PMID: 33584695 Free PMC article. Review.
-
Probing early tumor response to radiation therapy using hyperpolarized [1-¹³C]pyruvate in MDA-MB-231 xenografts.PLoS One. 2013;8(2):e56551. doi: 10.1371/journal.pone.0056551. Epub 2013 Feb 12. PLoS One. 2013. PMID: 23424666 Free PMC article.
-
Overview and considerations in bottom-up proteomics.Analyst. 2023 Jan 31;148(3):475-486. doi: 10.1039/d2an01246d. Analyst. 2023. PMID: 36383138 Free PMC article. Review.
References
-
- Bonafe N, Gilmore-Hebert M, Folk NL, Azodi M, Zhou Y, Chambers SK. Glyceraldehyde-3-phosphate dehydrogenase binds to the AU-rich 3′untranslated region of colony-stimulating factor-1 (CSF-1) messenger RNA in human ovarian cancer cells: possible role of CSF-1 posttranscriptional regulation and tumor phenotype. Cancer Res. 2005;65:3762–3771. - PubMed
-
- Brown VM, Krynetski EJ, Krynetskaia NF, Grieger D, Mukatira ST, Murti KG, Slaughter CA, Park HW, Evans WE. A novel CRM1-mediated nuclear export signal governs nuclear accumulation of glyceraldehyde-3-phosphate dehydrogenase following genotoxic stress. J Biol Chem. 2004;279:5984–5992. - PubMed
-
- Bruns GAP, Gerald PS. Human glyceraldehyde-3-phosphate dehydrogenase in man-rodent somatic cell hybrids. Science. 1976;192:54–56. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials