Identification and removal of nitroxide spin label contaminant: impact on PRE studies of α-helical membrane proteins in detergent
- PMID: 22389096
- PMCID: PMC3375759
- DOI: 10.1002/pro.2038
Identification and removal of nitroxide spin label contaminant: impact on PRE studies of α-helical membrane proteins in detergent
Abstract
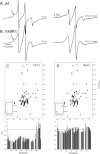
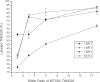
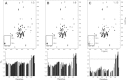
NMR paramagnetic relaxation enhancement (PRE) provides long-range distance constraints (~15-25 Å) that can be critical to determining overall protein topology, especially where long-range NOE information is unavailable such as in the case of larger proteins that require deuteration. However, several challenges currently limit the use of NMR PRE for α-helical membrane proteins. One challenge is the nonspecific association of the nitroxide spin label to the protein-detergent complex that can result in spurious PRE derived distance restraints. The effect of the nitroxide spin label contaminant is evaluated and quantified and a robust method for the removal of the contaminant is provided to advance the application of PRE restraints to membrane protein NMR structure determination.
Copyright © 2012 The Protein Society.
Figures
Similar articles
-
Structural origins of nitroxide side chain dynamics on membrane protein α-helical sites.Biochemistry. 2010 Nov 30;49(47):10045-60. doi: 10.1021/bi101148w. Epub 2010 Nov 8. Biochemistry. 2010. PMID: 20964375 Free PMC article.
-
Solution NMR Structure Determination of Polytopic α-Helical Membrane Proteins: A Guide to Spin Label Paramagnetic Relaxation Enhancement Restraints.Methods Enzymol. 2015;557:329-48. doi: 10.1016/bs.mie.2014.12.005. Epub 2015 Mar 17. Methods Enzymol. 2015. PMID: 25950972 Review.
-
Site-directed parallel spin-labeling and paramagnetic relaxation enhancement in structure determination of membrane proteins by solution NMR spectroscopy.J Am Chem Soc. 2006 Apr 5;128(13):4389-97. doi: 10.1021/ja0574825. J Am Chem Soc. 2006. PMID: 16569016 Free PMC article.
-
Expression, purification, and characterization of Thermotoga maritima membrane proteins for structure determination.Protein Sci. 2006 May;15(5):961-75. doi: 10.1110/ps.051874706. Epub 2006 Apr 5. Protein Sci. 2006. PMID: 16597824 Free PMC article.
-
Nitroxide spin-spin interactions: applications to protein structure and dynamics.Annu Rev Biophys Biomol Struct. 1999;28:129-53. doi: 10.1146/annurev.biophys.28.1.129. Annu Rev Biophys Biomol Struct. 1999. PMID: 10410798 Review.
Cited by
-
Optimization of Detergent-Mediated Reconstitution of Influenza A M2 Protein into Proteoliposomes.Membranes (Basel). 2018 Nov 8;8(4):103. doi: 10.3390/membranes8040103. Membranes (Basel). 2018. PMID: 30413063 Free PMC article.
-
Mapping membrane protein backbone dynamics: a comparison of site-directed spin labeling with NMR 15N-relaxation measurements.Biophys J. 2014 Oct 7;107(7):1697-702. doi: 10.1016/j.bpj.2014.08.018. Biophys J. 2014. PMID: 25296323 Free PMC article.
-
Backbone ¹H, ¹³C and ¹⁵N resonance assignments of the α-helical membrane protein TM0026 from Thermotoga maritima.Biomol NMR Assign. 2013 Oct;7(2):203-6. doi: 10.1007/s12104-012-9410-1. Epub 2012 Jul 28. Biomol NMR Assign. 2013. PMID: 23011877 Free PMC article.
References
-
- Nietlispach D, Gautier A. Solution NMR studies of polytopic alpha-helical membrane proteins. Curr Opin Struct Biol. 2011;21:497–508. - PubMed
-
- Patching SG. NMR structures of polytopic integral membrane proteins. Mol Membr Biol. 2011;28:370–397. - PubMed
-
- Sanders CR, Sonnichsen F. Solution NMR of membrane proteins: practice and challenges. Magn Reson Chem. 2006;44:S24–40. - PubMed
-
- Tamm LK, Liang B. NMR of membrane proteins in solution. Prog Nucl Magn Reson Spectrosc. 2006;48:201–210.
-
- Fernandez C, Wider G. TROSY in NMR studies of the structure and function of large biological macromolecules. Curr Opin Struct Biol. 2003;13:570–580. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

