Intramolecular fluorescence resonance energy transfer (FRET) sensors of the orexin OX1 and OX2 receptors identify slow kinetics of agonist activation
- PMID: 22389503
- PMCID: PMC3340242
- DOI: 10.1074/jbc.M111.334300
Intramolecular fluorescence resonance energy transfer (FRET) sensors of the orexin OX1 and OX2 receptors identify slow kinetics of agonist activation
Abstract
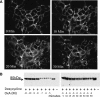
Intramolecular fluorescence resonance energy transfer (FRET) sensors able to detect changes in distance or orientation between the 3rd intracellular loop and C-terminal tail of the human orexin OX(1) and OX(2) G protein-coupled receptors following binding of agonist ligands were produced and expressed stably. These were directed to the plasma membrane and, despite the substantial sequence alterations introduced, in each case were able to elevate [Ca(2+)](i), promote phosphorylation of the ERK1/2 MAP kinases and become internalized effectively upon addition of the native orexin peptides. Detailed characterization of the OX(1) sensor demonstrated that it was activated with rank order of potency orexin A > orexin B > orexin A 16-33, that it bound antagonist ligands with affinity similar to the wild-type receptor, and that mutation of a single residue, D203A, greatly reduced the binding and function of orexin A but not antagonist ligands. Addition of orexin A to individual cells expressing an OX(1) sensor resulted in a time- and concentration-dependent reduction in FRET signal consistent with mass-action and potency/affinity estimates for the peptide. Compared with the response kinetics of a muscarinic M(3) acetylcholine receptor sensor upon addition of agonist, response of the OX(1) and OX(2) sensors to orexin A was slow, consistent with a multistep binding and activation process. Such sensors provide means to assess the kinetics of receptor activation and how this may be altered by mutation and sequence variation of the receptors.
Figures




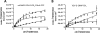
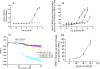

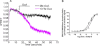
Similar articles
-
A role for PKD1 and PKD3 activation in modulation of calcium oscillations induced by orexin receptor 1 stimulation.Biochim Biophys Acta. 2010 Oct;1803(10):1206-12. doi: 10.1016/j.bbamcr.2010.07.001. Epub 2010 Jul 16. Biochim Biophys Acta. 2010. PMID: 20621130
-
Dynein light chain Tctex-type 1 modulates orexin signaling through its interaction with orexin 1 receptor.PLoS One. 2011;6(10):e26430. doi: 10.1371/journal.pone.0026430. Epub 2011 Oct 20. PLoS One. 2011. PMID: 22028875 Free PMC article.
-
Distinct recognition of OX1 and OX2 receptors by orexin peptides.J Pharmacol Exp Ther. 2003 May;305(2):507-14. doi: 10.1124/jpet.102.048025. Epub 2003 Jan 24. J Pharmacol Exp Ther. 2003. PMID: 12606634
-
Orexins/hypocretins: pain regulation and cellular actions.Curr Pharm Des. 2010;16(28):3089-100. doi: 10.2174/138161210793292483. Curr Pharm Des. 2010. PMID: 20687883 Review.
-
Targeting orexin receptors: Recent advances in the development of subtype selective or dual ligands for the treatment of neuropsychiatric disorders.Med Res Rev. 2023 Sep;43(5):1607-1667. doi: 10.1002/med.21959. Epub 2023 Apr 10. Med Res Rev. 2023. PMID: 37036052 Review.
Cited by
-
Diurnal inhibition of NMDA-EPSCs at rat hippocampal mossy fibre synapses through orexin-2 receptors.J Physiol. 2014 Oct 1;592(19):4277-95. doi: 10.1113/jphysiol.2014.272757. Epub 2014 Aug 1. J Physiol. 2014. PMID: 25085886 Free PMC article.
-
[Orexin-A inhibits γ-aminobutyric acid current of neonatal rat spinal cord ventral horn neurons by activating OX1R, OX2R and Ca2+-independent PKC].Nan Fang Yi Ke Da Xue Xue Bao. 2021 May 20;41(5):694-701. doi: 10.12122/j.issn.1673-4254.2021.05.09. Nan Fang Yi Ke Da Xue Xue Bao. 2021. PMID: 34134956 Free PMC article. Chinese.
-
Optical probes based on G protein-coupled receptors - added work or added value?Br J Pharmacol. 2016 Jan;173(2):255-66. doi: 10.1111/bph.13382. Epub 2015 Dec 19. Br J Pharmacol. 2016. PMID: 26562218 Free PMC article. Review.
-
A Novel 5-HT1B Receptor Agonist of Herbal Compounds and One of the Therapeutic Uses for Alzheimer's Disease.Front Pharmacol. 2021 Sep 6;12:735876. doi: 10.3389/fphar.2021.735876. eCollection 2021. Front Pharmacol. 2021. PMID: 34552493 Free PMC article.
-
Neural circuit interactions between the dorsal raphe nucleus and the lateral hypothalamus: an experimental and computational study.PLoS One. 2014 Feb 6;9(2):e88003. doi: 10.1371/journal.pone.0088003. eCollection 2014. PLoS One. 2014. PMID: 24516577 Free PMC article.
References
-
- Sakurai T., Mieda M. (2011) Connectomics of orexin-producing neurons: interface of systems of emotion, energy homeostasis and arousal. Trends Pharmacol. Sci. 32, 451–462 - PubMed
-
- Chiou L. C., Lee H. J., Ho Y. C., Chen S. P., Liao Y. Y., Ma C. H., Fan P. C., Fuh J. L., Wang S. J. (2010) Orexins/hypocretins: pain regulation and cellular actions. Curr. Pharm. Des. 16, 3089–3100 - PubMed
-
- Ritchie C., Okuro M., Kanbayashi T., Nishino S. (2010) Hypocretin ligand deficiency in narcolepsy: recent basic and clinical insights. Curr. Neurol. Neurosci. Rep. 10, 180–189 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

