Determination of the size distribution of blood microparticles directly in plasma using atomic force microscopy and microfluidics
- PMID: 22391880
- PMCID: PMC3388260
- DOI: 10.1007/s10544-012-9642-y
Determination of the size distribution of blood microparticles directly in plasma using atomic force microscopy and microfluidics
Abstract
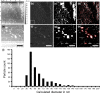
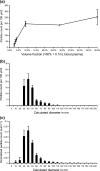
Microparticles, also known as microvesicles, found in blood plasma, urine, and most other body fluids, may serve as valuable biomarkers of diseases such as cardiovascular diseases, systemic inflammatory disease, thrombosis, and cancer. Unfortunately, the detection and quantification of microparticles are hampered by the microscopic size of these particles and their relatively low abundance in blood plasma. The use of a combination of microfluidics and atomic force microscopy to detect microparticles in blood plasma circumvents both problems. In this study, capture of a specific subset of microparticles directly from blood plasma on antibody-coated mica surface is demonstrated. The described method excludes isolation and washing steps to prepare microparticles, improves the detection sensitivity, and yields the size distribution of the captured particles. The majority of the captured particles have a size ranging from 30 to 90 nm, which is in good agreement with prior results obtained with microparticles immediately isolated from fresh plasma. Furthermore, the qualitative shape of the size distribution of microparticles is shown not to be affected by high-speed centrifugation or the use of the microfluidic circuit, demonstrating the relative stable nature of microparticles ex vivo.
Figures


References
-
- Burnier L, Fontana P, Kwak BR, Angelillo-Scherrer A. Thromb. Haemost. 2009;101:439. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

