Interaction of cowpea mosaic virus nanoparticles with surface vimentin and inflammatory cells in atherosclerotic lesions
- PMID: 22394183
- PMCID: PMC3567616
- DOI: 10.2217/nnm.11.185
Interaction of cowpea mosaic virus nanoparticles with surface vimentin and inflammatory cells in atherosclerotic lesions
Abstract
Aims: Detection of atherosclerosis has generally been limited to the late stages of development, after cardiovascular symptoms present or a clinical event occurs. One possibility for early detection is the use of functionalized nanoparticles. The aim of this study was the early imaging of atherosclerosis using nanoparticles with a natural affinity for inflammatory cells in the lesion.
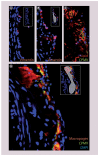
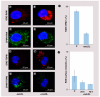
Materials & methods: We investigated uptake of cowpea mosaic virus by macrophages and foam cells in vitro and correlated this with vimentin expression. We also examined the ability of cowpea mosaic virus to interact with atherosclerotic lesions in a murine model of atherosclerosis.
Results & conclusion: We found that uptake of cowpea mosaic virus is increased in areas of atherosclerotic lesion. This correlated with increased surface vimentin in the lesion compared with nonlesion vasculature. In conclusion, cowpea mosaic virus and its vimentin-binding region holds potential for use as a targeting ligand for early atherosclerotic lesions, and as a probe for detecting upregulation of surface vimentin during inflammation.
Figures



References
-
- Slevin M, Badimon L, Grau-Olivares M, et al. Combining nanotechnology with current biomedical knowledge for the vascular imaging and treatment of atherosclerosis. Mol. Biosyst. 2010;6(3):444–450. - PubMed
-
- Cybulsky MI, Jongstra-Bilen J. Resident intimal dendritic cells and the initiation of atherosclerosis. Curr. Opin. Lipidol. 2010;21(5):397–403. - PubMed
-
- Tavora F, Cresswell N, Li L, Ripple M, Burke A. Immunolocalisation of fibrin in coronary atherosclerosis: implications for necrotic core development. Pathology. 2010;42(1):15–22. - PubMed
-
- Ovchinnikova O, Gylfe A, Bailey L, et al. Osteoprotegerin promotes fibrous cap formation in atherosclerotic lesions of ApoE-deficient mice--brief report. Arterioscler. Thromb. Vasc. Biol. 2009;29(10):1478–1480. - PubMed
Websites
-
- ImageJ: Image processing and analysis in Java. http://rsbweb.nih.gov/ij/
-
- Imaris (bitplane) software. www.bitplane.com/go/products/imaris.
-
- Virus particle explorer. http://viperDB.scripps.edu.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
