Serum proteomic profiling in patients with drug-induced liver injury
- PMID: 22403816
- PMCID: PMC3654532
- DOI: 10.1111/j.1365-2036.2011.04982.x
Serum proteomic profiling in patients with drug-induced liver injury
Abstract
Background: Idiosyncratic drug-induced liver injury (DILI) is a complex disorder that is difficult to predict, diagnose and treat.
Aim: To describe the global serum proteome of patients with DILI and controls.
Methods: A label-free, mass spectrometry-based quantitative proteomic approach was used to explore protein expression in serum samples from 74 DILI patients (collected within 14 days of DILI onset) and 40 controls. A longitudinal analysis was conducted in a subset of 21 DILI patients with available 6-month follow-up serum samples.
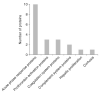
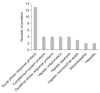
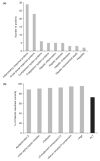
Results: Comparison of DILI patients based on pattern, severity and causality assessment of liver injury revealed many differentially expressed priority 1 proteins among groups. Expression of fumarylacetoacetase was correlated with alanine aminotransferase (ALT; r = 0.237; P = 0.047), aspartate aminotransferase (AST; r = 0.389; P = 0.001) and alkaline phosphatase (r = -0.240; P = 0.043), and this was the only protein with significant differential expression when comparing patients with hepatocellular vs. cholestatic or mixed injury. In the longitudinal analysis, expression of 53 priority 1 proteins changed significantly from onset of DILI to 6-month follow-up, and nearly all proteins returned to expression levels comparable to control subjects. Ninety-two serum priority 1 proteins with significant differential expression were identified when comparing the DILI and control groups. Pattern analysis revealed proteins that are components of inflammation, immune system activation and several hepatotoxicity-specific pathways. Apolipoprotein E expression had the greatest power to differentiate DILI patients from controls (89% correct classification; AUROC = 0.97).
Conclusion: This proteomic analysis identified differentially expressed proteins that are components of pathways previously implicated in the pathogenesis of idiosyncratic drug-induced liver injury.
Figures
Comment in
-
Commentary: proteomics and drug-induced liver injury.Aliment Pharmacol Ther. 2012 Mar;35(6):740-1. doi: 10.1111/j.1365-2036.2012.04999.x. Aliment Pharmacol Ther. 2012. PMID: 22348521 No abstract available.
Similar articles
-
The value of serum aspartate aminotransferase and gamma-glutamyl transpetidase as biomarkers in hepatotoxicity.Liver Int. 2015 Nov;35(11):2474-82. doi: 10.1111/liv.12834. Epub 2015 Apr 8. Liver Int. 2015. PMID: 25809419
-
Characteristics of idiosyncratic drug-induced liver injury in children: results from the DILIN prospective study.J Pediatr Gastroenterol Nutr. 2011 Aug;53(2):182-9. doi: 10.1097/MPG.0b013e31821d6cfd. J Pediatr Gastroenterol Nutr. 2011. PMID: 21788760 Free PMC article.
-
Persistent liver biochemistry abnormalities are more common in older patients and those with cholestatic drug induced liver injury.Am J Gastroenterol. 2015 Oct;110(10):1450-9. doi: 10.1038/ajg.2015.283. Epub 2015 Sep 8. Am J Gastroenterol. 2015. PMID: 26346867 Free PMC article.
-
Drug-induced liver injury.Mayo Clin Proc. 2014 Jan;89(1):95-106. doi: 10.1016/j.mayocp.2013.09.016. Mayo Clin Proc. 2014. PMID: 24388027 Review.
-
The application of cytokeratin-18 as a biomarker for drug-induced liver injury.Arch Toxicol. 2021 Nov;95(11):3435-3448. doi: 10.1007/s00204-021-03121-0. Epub 2021 Jul 29. Arch Toxicol. 2021. PMID: 34322741 Free PMC article. Review.
Cited by
-
Advances in drug-induced liver injury research: in vitro models, mechanisms, omics and gene modulation techniques.Cell Biosci. 2024 Nov 2;14(1):134. doi: 10.1186/s13578-024-01317-2. Cell Biosci. 2024. PMID: 39488681 Free PMC article. Review.
-
The Protective Effect of Sonneratia apetala Fruit Extract on Acetaminophen-Induced Liver Injury in Mice.Evid Based Complement Alternat Med. 2019 Jun 19;2019:6919834. doi: 10.1155/2019/6919834. eCollection 2019. Evid Based Complement Alternat Med. 2019. PMID: 31320915 Free PMC article.
-
Pathogenesis of idiosyncratic drug-induced liver injury and clinical perspectives.Gastroenterology. 2014 Apr;146(4):914-28. doi: 10.1053/j.gastro.2013.12.032. Epub 2013 Dec 31. Gastroenterology. 2014. PMID: 24389305 Free PMC article. Review.
-
Association between apolipoprotein E genotype, chronic liver disease, and hepatitis B virus.Clin Mol Hepatol. 2012 Sep;18(3):295-301. doi: 10.3350/cmh.2012.18.3.295. Epub 2012 Sep 25. Clin Mol Hepatol. 2012. PMID: 23091810 Free PMC article.
-
Profiles of serum cytokines in acute drug-induced liver injury and their prognostic significance.PLoS One. 2013 Dec 27;8(12):e81974. doi: 10.1371/journal.pone.0081974. eCollection 2013. PLoS One. 2013. PMID: 24386086 Free PMC article.
References
-
- Navarro VJ, Senior JR. Drug-related hepatotoxicity. N Engl J Med. 2006;354:731–9. - PubMed
-
- Watkins PB, Seeff LB. Drug-induced liver injury: summary of a single topic clinical research conference. Hepatology. 2006;43:618–31. - PubMed
-
- Abboud G, Kaplowitz N. Drug-induced liver injury. Drug Saf. 2007;30:277–94. - PubMed
-
- Bonkovsky HL, Shedlofsky SI, Jones DP, et al. Drug-induced liver injury. In: Boyer TD, Manns MP, Sanyal A, editors. Zakim and Boyer’s Hepatology – A Textbook of Liver Disease. 6th ed Saunders-Elsevier; Philadelphia: 2011.
-
- Ostapowicz G, Fontana RJ, Schiodt FV, et al. Results of a prospective study of acute liver failure at 17 tertiary care centers in the United States. Ann Intern Med. 2002;137:947–54. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 1U01DK065238/DK/NIDDK NIH HHS/United States
- 1U01DK065021/DK/NIDDK NIH HHS/United States
- U01DK065193/DK/NIDDK NIH HHS/United States
- U01 DK065201/DK/NIDDK NIH HHS/United States
- U01 DK065238/DK/NIDDK NIH HHS/United States
- U01 DK065193/DK/NIDDK NIH HHS/United States
- 1U01DK065201/DK/NIDDK NIH HHS/United States
- U01 DK065176/DK/NIDDK NIH HHS/United States
- 1U01DK065176/DK/NIDDK NIH HHS/United States
- U01 DK065211/DK/NIDDK NIH HHS/United States
- K24 DK069290/DK/NIDDK NIH HHS/United States
- 1U01DK065193/DK/NIDDK NIH HHS/United States
- 1U01DK065211/DK/NIDDK NIH HHS/United States
- 3 U01 DK065211-0751-ARRA/DK/NIDDK NIH HHS/United States
- 1U01DK065184/DK/NIDDK NIH HHS/United States
- U01 DK065184/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
