Filamins in mechanosensing and signaling
- PMID: 22404683
- PMCID: PMC5508560
- DOI: 10.1146/annurev-biophys-050511-102252
Filamins in mechanosensing and signaling
Abstract
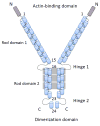
Filamins are essential, evolutionarily conserved, modular, multidomain, actin-binding proteins that organize the actin cytoskeleton and maintain extracellular matrix connections by anchoring actin filaments to transmembrane receptors. By cross-linking and anchoring actin filaments, filamins stabilize the plasma membrane, provide cellular cortical rigidity, and contribute to the mechanical stability of the plasma membrane and the cell cortex. In addition to binding actin, filamins interact with more than 90 other binding partners including intracellular signaling molecules, receptors, ion channels, transcription factors, and cytoskeletal and adhesion proteins. Thus, filamins scaffold a wide range of signaling pathways and are implicated in the regulation of a diverse array of cellular functions including motility, maintenance of cell shape, and differentiation. Here, we review emerging structural and functional evidence that filamins are mechanosensors and/or mechanotransducers playing essential roles in helping cells detect and respond to physical forces in their local environment.
Figures


References
-
- Barry CP, Xie J, Lemmon V, Young AP. Molecular characterization of a multi-promoter gene encoding a chicken filamin protein. J Biol Chem. 1993;268:25577–86. - PubMed
-
- Bellanger JM, Astier C, Sardet C, Ohta Y, Stossel TP, Debant A. The Rac1- and RhoG-specific GEF domain of Trio targets filamin to remodel cytoskeletal actin. Nat Cell Biol. 2000;2:888–92. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

